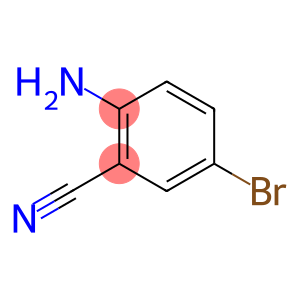
2-Amino-5-bromobenzonitrile
2-Amino-5-bromobenzonitrile
CAS: 39263-32-6
Molecular Formula: C7H5BrN2
2-Amino-5-bromobenzonitrile - Names and Identifiers
| Name | 2-Amino-5-bromobenzonitrile |
| Synonyms | 4-Bromo-2-cyanoaniline 5-Bromoanthranilonitrile 2-Amino-5-brombenzonitrile 2-amino-5-bromobenonitrile 2-AMINO-5-BROMBENZONITRILE 2-AMINO-5-BROMOBENZONITRILE 2-Amino-5-bromobenzonitrile 2-AMINO-5-BROMOBENZENECARBONITRILE |
| CAS | 39263-32-6 |
| EINECS | 254-387-8 |
| InChI | InChI=1/C7H5BrN2/c8-6-1-2-7(10)5(3-6)4-9/h1-3H,10H2 |
2-Amino-5-bromobenzonitrile - Physico-chemical Properties
| Molecular Formula | C7H5BrN2 |
| Molar Mass | 197.03 |
| Density | 1.6480 (rough estimate) |
| Melting Point | 96-100°C(lit.) |
| Boling Point | 288.4±25.0 °C(Predicted) |
| Flash Point | 128.2°C |
| Solubility | Soluble in methanol. |
| Vapor Presure | 0.00235mmHg at 25°C |
| Appearance | White to light white crystals |
| Color | Off-white to pale brown |
| pKa | 1.11±0.10(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Refractive Index | 1.5500 (estimate) |
| MDL | MFCD00158946 |
2-Amino-5-bromobenzonitrile - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R43 - May cause sensitization by skin contact R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S36/37 - Wear suitable protective clothing and gloves. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S22 - Do not breathe dust. |
| UN IDs | 3439 |
| WGK Germany | 3 |
| HS Code | 29269090 |
| Hazard Note | Irritant |
| Hazard Class | 6.1 |
| Packing Group | III |
2-Amino-5-bromobenzonitrile - Reference Information
| introduction | 2-amino -5-bromobenzonitrile is a yellow-brown or off-white solid with good solubility in methanol. 2-Amino-5-bromobenzonitrile is a nitrile derivative, which is an intermediate for the synthesis of pesticides, aliphatic amines, benzoic acids and advanced coatings. |
| Uses | 2-amino-5-bromobenzonitrile can be used as a pharmaceutical intermediate, a synthetic intermediate for coatings, and also in the synthesis of polymer materials There are applications. In the synthetic conversion, the bromine unit on the benzene ring can be introduced into a series of aryl groups or alkyl groups through the Suzuki coupling reaction, and can also be converted into boric acid units for subsequent conversion; the cyano group on the benzene ring can be easily converted into aldehyde groups, carboxyl groups, amides and other functional groups. |
| synthesis method | the AcOH(120ml) solution of ammonium bromide (10.3g,0.105mol), hydrogen peroxide (10.2ml,35% aqueous solution, 0.105mol),2-aminobenzonitrile (11.8g,0.1mol) was added to the reaction bottle in batches, the mixture was stirred at room temperature for about 24 hours until LCMS analysis showed that the reaction was complete. Concentrate the mixture to remove AcOH, then stir the residue with 30% aqueous solution of NaOH until alkaline, filter to remove the resulting solid and wash with water before drying, and then dissolve the solid in excess DCM. Concentrate the solution until precipitation begins, and then let it stand until the crystallization is complete. The obtained solid was filtered to remove and washed with a small amount of DCM to obtain 2-amino-5-bromobenzonitrile, an off-white crystalline solid (19.2g,97%). |
Last Update:2024-04-09 02:00:08
Supplier List
Product Name: 2-AMINO-5-BROMOBENZONITRILE Request for quotation
CAS: 39263-32-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 39263-32-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2-Amino-5-bromobenzonitrile Visit Supplier Webpage Request for quotationCAS: 39263-32-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 2-Amino-5-bromobenzonitrile Visit Supplier Webpage Request for quotationCAS: 39263-32-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 2-Amino-5-bromobenzonitrile Visit Supplier Webpage Request for quotation
CAS: 39263-32-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 39263-32-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2-AMINO-5-BROMOBENZONITRILE Request for quotation
CAS: 39263-32-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 39263-32-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2-Amino-5-bromobenzonitrile Visit Supplier Webpage Request for quotationCAS: 39263-32-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 2-Amino-5-bromobenzonitrile Visit Supplier Webpage Request for quotationCAS: 39263-32-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 2-Amino-5-bromobenzonitrile Visit Supplier Webpage Request for quotation
CAS: 39263-32-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 39263-32-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



