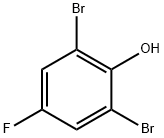
2,6-Dibromo-4-fluorophenol
2,6-Dibromo-4-fluorophenol
CAS: 344-20-7
Molecular Formula: C6H3Br2FO
2,6-Dibromo-4-fluorophenol - Names and Identifiers
| Name | 2,6-Dibromo-4-fluorophenol |
| Synonyms | 2,6-Dibromo-4-fluorophenol 4-Fluoro-2,6-dibromophenol 2,6-DIBROMO-4-FLUOROPHENOL 2,6-Dibromo-4-2-fluorophenol 1-Bromo-3,4,5-trifluorobezene |
| CAS | 344-20-7 |
| EINECS | 206-451-1 |
| InChI | InChI=1/C6H3Br2FO/c7-4-1-3(9)2-5(8)6(4)10/h1-2,10H |
| InChIKey | RRAZCUUOWIDAJS-UHFFFAOYSA-N |
2,6-Dibromo-4-fluorophenol - Physico-chemical Properties
| Molecular Formula | C6H3Br2FO |
| Molar Mass | 269.89 |
| Density | 2.168±0.06 g/cm3(Predicted) |
| Melting Point | 55-57°C |
| Boling Point | 219.5±35.0 °C(Predicted) |
| Flash Point | 86.5°C |
| Vapor Presure | 0.0807mmHg at 25°C |
| BRN | 1868033 |
| pKa | 6.92±0.23(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.616 |
2,6-Dibromo-4-fluorophenol - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S36 - Wear suitable protective clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| Hazard Note | Harmful |
2,6-Dibromo-4-fluorophenol - Reference Information
| properties | white to light yellow crystal powder |
| use | 2, 6-dibromo-4-fluorophenol is an important intermediate for the preparation of α-amino acid derivatives and antispasmodic drugs. Halogenated phenol compounds are important organic synthesis and pharmaceutical intermediates, which are widely used in synthetic medicine, pesticides, plastic and rubber additives, disinfectants and preservatives and other fine chemicals. Interhalophenol compounds are the raw materials for the synthesis of non-morphine powerful analgesics tramadol hydrochloride, triarylmethane thiophene anti-tuberculosis drugs and 4-arylpiperidine anti-itching drugs. 2, 4-dibromophenol (DBP) is an important organic chemical raw material, which is widely used in epoxy resin, phenolic resin, polyurethane and other reactive flame retardants and intermediates in the synthesis of other chemical raw materials. |
| Preparation | At present, the synthesis method of 2, 6-dibromo-4-fluorophenol is mainly based on 4-fluorophenol as raw material, and the target compound is obtained by bromination reaction [1]. Or use 3, 5-dibromo-1-fluorobenzene as the starting material to obtain the target compound 2, 6-dibromo-4-fluorophenol through two-step reaction of boration and oxidation. The synthesis reaction formula is as follows: Figure 12, 6-dibromo -4-fluorophenol synthesis reaction formula 2,6-dibromo -1-fluorobenzene boronic acid preparation in 250 mL three-mouth bottle Add 2,2,6, 6-tetramethylpiperidine (10. 0g,70. 8mmol) and 160 mL dry tetrahydrofuran, under the protection of N 2, in a low-temperature reactor to -78 ℃, slowly add n-butyl lithium (21. 2 mL,53. 0 mmol), after dropping, stir for 30 min, slowly add 3,5-dibromo -1-fluorobenzene (8. 3g,35. 2 mmol), after dropping, stir for 1h, and then slowly add triisopropyl borate (10. 0g,53. 0 mmol). After dropping, continue stirring for 2 h to complete the reaction. The reaction solution was slowly poured into ice water, extracted with ethyl acetate (50 mL × 3) to extract impurities, the aqueous phase was adjusted to pH = 3~4 with 1 mol · L-1 hydrochloric acid, the solid was precipitated, filtered, dried, soaked and pulped with 10 mL n-hexane, filtered and dried to obtain 5.8g 2, 6-dibromo-1-fluorophenylboronic acid. Preparation of 2,6-Dibromo -4-Fluorophenol Add 40 g of water and potassium hydroxide (1. 2g,21. 4mmol) into a 250 mL single-mouth bottle, after complete dissolution, add 2,6-dibromo -1-fluorophenylboronic acid (3g,10. 7 mmol), slowly add 30% aqueous hydrogen peroxide solution (6. 07g,53. 5 mmol), after dropping, continue to stir for 10 min, after the reaction, adjust to pH = 5~6 with 1 mol · L-1 hydrochloric acid, extract with ethyl acetate (80 mL× 2), combine organic phases, dry with anhydrous sodium sulfate, evaporate to dry to obtain light yellow solid crude product, soak and beat with 5 mL n-hexane, filter and dry to obtain 2.6 g2, 6-dibromo-4-fluorophenol. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 2,6-DIBROMO-4-FLUOROPHENOL Request for quotation
CAS: 344-20-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 344-20-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2,6-Dibromo-4-fluorophenol Visit Supplier Webpage Request for quotationCAS: 344-20-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,6-DIBROMO-4-FLUOROPHENOL Request for quotation
CAS: 344-20-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 344-20-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2,6-Dibromo-4-fluorophenol Visit Supplier Webpage Request for quotationCAS: 344-20-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
View History

