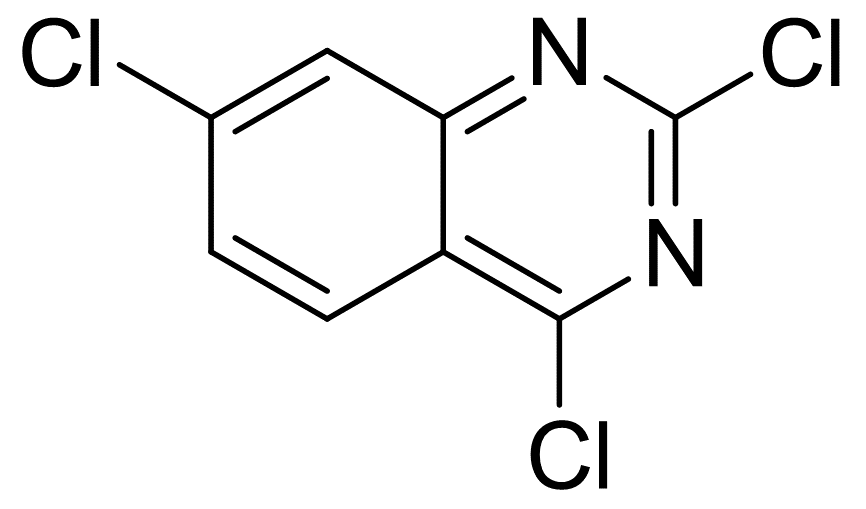
2,4,7-trichloroquinazoline
2,4,7-trichloroquinazoline
CAS: 6625-94-1
Molecular Formula: C8H3Cl3N2
2,4,7-trichloroquinazoline - Names and Identifiers
| Name | 2,4,7-trichloroquinazoline |
| Synonyms | 2,4,7-TRICHLOROQUINAZOLINE 2,4,7-trichloroquinazoline 2,4,7-Trichloroquinazoline Quinazoline, 2,4,7-trichloro- 2,4-Dichloro-7-chloroquinazoline |
| CAS | 6625-94-1 |
| InChI | InChI=1/C8H3Cl3N2/c9-4-1-2-5-6(3-4)12-8(11)13-7(5)10/h1-3H |
2,4,7-trichloroquinazoline - Physico-chemical Properties
| Molecular Formula | C8H3Cl3N2 |
| Molar Mass | 233.48 |
| Density | 1.600±0.06 g/cm3(Predicted) |
| Melting Point | 127 °C |
| Boling Point | 309.1±24.0 °C(Predicted) |
| Flash Point | 169.6°C |
| Vapor Presure | 0.00119mmHg at 25°C |
| pKa | -1.14±0.30(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.677 |
2,4,7-trichloroquinazoline - Introduction
2,4, 7-tricholoquinazoline is an organic compound with the chemical formula C9H4Cl3N. The following is an introduction to its nature, use, formulation and safety information:
Nature:
-Appearance: Colorless to light yellow solid.
-Melting point: about 110-112°C.
-Boiling point: about 310°C.
-Solubility: Soluble in organic solvents, almost insoluble in water.
-Chemical stability: relatively stable at room temperature, but may decompose under the influence of light and heat.
Use:
- 2,4,7-trichloroquinazoline is an important intermediate compound that can be used to synthesize other organic compounds.
-It can also be used as a precursor for antibacterial agents, antiviral drugs and pesticides.
Preparation Method:
- 2,4,7-trichloroquinazoline can be synthesized by a variety of methods, one of the common methods is to take 2,4-dichlorobenzene ammonia as the starting material, in the presence of a chlorinating agent through a series of reaction steps to obtain the target product.
Safety Information:
- 2,4,7-trichloroquinazoline is an organic compound and should be handled with caution to avoid contact with skin and inhalation of dust.
-During operation, storage and transportation, appropriate safety measures need to be taken, including wearing protective gloves, respirators, etc.
-Due to its chemical properties and potential toxicity, it may have a negative impact on the environment, so the corresponding environmental protection regulations and standards should be followed during use and handling.
Nature:
-Appearance: Colorless to light yellow solid.
-Melting point: about 110-112°C.
-Boiling point: about 310°C.
-Solubility: Soluble in organic solvents, almost insoluble in water.
-Chemical stability: relatively stable at room temperature, but may decompose under the influence of light and heat.
Use:
- 2,4,7-trichloroquinazoline is an important intermediate compound that can be used to synthesize other organic compounds.
-It can also be used as a precursor for antibacterial agents, antiviral drugs and pesticides.
Preparation Method:
- 2,4,7-trichloroquinazoline can be synthesized by a variety of methods, one of the common methods is to take 2,4-dichlorobenzene ammonia as the starting material, in the presence of a chlorinating agent through a series of reaction steps to obtain the target product.
Safety Information:
- 2,4,7-trichloroquinazoline is an organic compound and should be handled with caution to avoid contact with skin and inhalation of dust.
-During operation, storage and transportation, appropriate safety measures need to be taken, including wearing protective gloves, respirators, etc.
-Due to its chemical properties and potential toxicity, it may have a negative impact on the environment, so the corresponding environmental protection regulations and standards should be followed during use and handling.
Last Update:2024-04-09 21:04:16
Supplier List
Product Name: 2,4,7-TRICHLOROQUINAZOLINE Request for quotation
CAS: 6625-94-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6625-94-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2,4,7-Trichloroquinazoline Visit Supplier Webpage Request for quotationCAS: 6625-94-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4,7-Trichloroquinazoline Visit Supplier Webpage Request for quotation
CAS: 6625-94-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6625-94-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2,4,7-TRICHLOROQUINAZOLINE Request for quotation
CAS: 6625-94-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6625-94-1
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2,4,7-Trichloroquinazoline Visit Supplier Webpage Request for quotationCAS: 6625-94-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4,7-Trichloroquinazoline Visit Supplier Webpage Request for quotation
CAS: 6625-94-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6625-94-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


