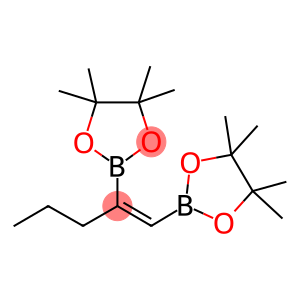
2,2'-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane)
2,2'-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane)
CAS: 307531-75-5
Molecular Formula: C17H32B2O4
2,2'-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) - Names and Identifiers
| Name | 2,2'-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) |
| Synonyms | Bis(pinacolcyclicester) (E)-1-Pentene-1,2-diboronicacid (E)-1-PENTENE-1,2-DIBORONIC ACID BIS(PINACOL) ESTER 1-cis-1,2-bis(4,4,5,5-tetramethyl-1,3,2-dioxoboralan-2-yl)pentene 1-CIS-1,2-BIS(4,4,5,5-TETRAMETHYL-1,3,2-DIOXABOROLAN-2-YL)PENTENE 1-cis-1,2-Bis(4,4,5,5-tetramethyl-1,3,2-dioxaboralan-2-yl)pentene 2,2'-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) 2,2'-[(1Z)-1-Propyl-1,2-ethenediyl]bis[4,4,5,5-tetramethyl-1,3,2-dioxaborolanel 2,2μ-[(1Z)-1-Propyl-1,2-ethenediyl]bis[4,4,5,5-tetramethyl-1,3,2-dioxaborolane], 1-[cis-1,2-Bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)] pentene |
| CAS | 307531-75-5 |
| EINECS | 000-000-0 |
| InChI | InChI=1/C17H32B2O4/c1-10-11-13(19-22-16(6,7)17(8,9)23-19)12-18-20-14(2,3)15(4,5)21-18/h12H,10-11H2,1-9H3/b13-12- |
2,2'-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) - Physico-chemical Properties
| Molecular Formula | C17H32B2O4 |
| Molar Mass | 322.06 |
| Density | 0.954g/mLat 25°C(lit.) |
| Melting Point | 301-303 |
| Boling Point | 301-303°C(lit.) |
| Flash Point | 169°F |
| Vapor Presure | 0.00175mmHg at 25°C |
| Refractive Index | n20/D 1.455(lit.) |
2,2'-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| UN IDs | NA 1993 / PGIII |
| WGK Germany | 3 |
| Hazard Note | Irritant |
2,2'-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) - Introduction
2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) is an organic compound with the chemical formula C30H56B2O4, the molecular weight is about 511.41G/mole. It is an organic boron compound with high spatial orientation.
2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane) is commonly used as a ligand, it can form stable coordination compounds with transition metals for catalytic reduction reactions, cross-coupling reactions and other organic synthesis reactions. It can be used as a synthetic intermediate of boron Ester in organic synthesis.
The preparation method of the compound can be carried out by the following steps:
1. First, 4,4, 5,5-tetramethyl -1,3, 2-dioxaboropentane (CAS number: 145637-88-0) and 2,2 '-(1E)-Pent-1-ene-1, 2-dimethylene diphenyl ether (CAS No.: 3944-80-1) was reacted. The reaction is stirred in an organic solvent, usually at a low temperature in an inert atmosphere.
2. Next, the resulting product is separated and purified by crystallization or column chromatography.
Regarding safety information, 2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane) may pose a potential risk to human health. When in use, should adopt strict laboratory practice, wear appropriate personal protective equipment (such as laboratory atmosphere type gas mask, chemical protective gloves, safety goggles, etc.). In addition, ensure that you operate in a well-ventilated area and avoid inhaling the gases, vapors or dust that are generated. In case of skin contact or accidental inhalation, seek medical attention immediately.
In addition, 2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) should be stored in a dry, cool, A well-ventilated area and away from fire and oxidants. Avoid contact with strong acid, strong alkali and other substances during use.
In General, 2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane) is an important organic boron compound, it is often used in organic synthesis and catalytic reactions, but it is necessary to pay attention to safe operation when using it.
2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane) is commonly used as a ligand, it can form stable coordination compounds with transition metals for catalytic reduction reactions, cross-coupling reactions and other organic synthesis reactions. It can be used as a synthetic intermediate of boron Ester in organic synthesis.
The preparation method of the compound can be carried out by the following steps:
1. First, 4,4, 5,5-tetramethyl -1,3, 2-dioxaboropentane (CAS number: 145637-88-0) and 2,2 '-(1E)-Pent-1-ene-1, 2-dimethylene diphenyl ether (CAS No.: 3944-80-1) was reacted. The reaction is stirred in an organic solvent, usually at a low temperature in an inert atmosphere.
2. Next, the resulting product is separated and purified by crystallization or column chromatography.
Regarding safety information, 2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane) may pose a potential risk to human health. When in use, should adopt strict laboratory practice, wear appropriate personal protective equipment (such as laboratory atmosphere type gas mask, chemical protective gloves, safety goggles, etc.). In addition, ensure that you operate in a well-ventilated area and avoid inhaling the gases, vapors or dust that are generated. In case of skin contact or accidental inhalation, seek medical attention immediately.
In addition, 2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) should be stored in a dry, cool, A well-ventilated area and away from fire and oxidants. Avoid contact with strong acid, strong alkali and other substances during use.
In General, 2,2 '-(1E)-pent-1-ene-1,2-diylbis(4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane) is an important organic boron compound, it is often used in organic synthesis and catalytic reactions, but it is necessary to pay attention to safe operation when using it.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 1-CIS-1,2-BIS(4,4,5,5-TETRAMETHYL-1,3,2-DIOXABOROLAN-2-YL)PENTENE Request for quotation
CAS: 307531-75-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 307531-75-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 1-CIS-1,2-BIS(4,4,5,5-TETRAMETHYL-1,3,2-DIOXABOROLAN-2-YL)PENTENE Request for quotation
CAS: 307531-75-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 307531-75-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
