198543-96-3
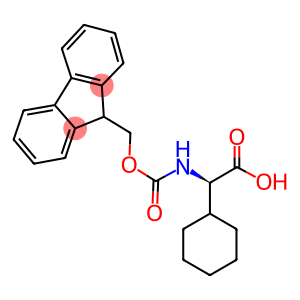
Fmoc-cyclohexyl-D-Gly-OH
CAS: 198543-96-3
Molecular Formula: C23H25NO4
198543-96-3 - Names and Identifiers
198543-96-3 - Physico-chemical Properties
| Molecular Formula | C23H25NO4 |
| Molar Mass | 379.45 |
| Density | 1.238 |
| Melting Point | 161-187℃ |
| Boling Point | 602.9±38.0 °C(Predicted) |
| Appearance | White to off-white powder |
| pKa | 3.89±0.10(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
198543-96-3 - Risk and Safety
| Hazard Class | IRRITANT |
198543-96-3 - Reference Information
| Use | Fluorene methoxycarbonyl D-cyclohexylglycine is a common pharmaceutical and chemical intermediate, fluorenyl chloride (FMOC-Cl) is a chloroformate. It is used to form fluorene methoxycarbonyl (FMOC group) into fluorene methoxycarbonyl carbamate FMOC carbamate, which is a common amino protecting group. |
| Preparation | The synthesis of fluorene methoxycarbonyl D-cyclohexylglycine has been reported in the literature. Using cyclohexyl bromide as the raw material, in the presence of sodium alkoxide, it undergoes a alkylation reaction with diethyl malonate, and then hydrolyzes, acidifies, and decarboxylates to obtain cyclohexyl acetic acid; the latter is then brominated and ammonolysis DL-Cyclohexylglycine; finally, acylation reaction occurs in the presence of fluorene methoxycarbonyl chloride to obtain fluorene methoxycarbonyl D-cyclohexylglycine [1]. The synthesis reaction route is as follows: |
Last Update:2024-04-09 15:16:48
Supplier List
Spot supply
Product Name: Fmoc-D-Chg-OH Visit Supplier Webpage Request for quotationCAS: 198543-96-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: FMOC-D-CHG-OH Request for quotation
CAS: 198543-96-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 198543-96-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: FMOC-D-CHG-OH Visit Supplier Webpage Request for quotation
CAS: 198543-96-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 198543-96-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Fmoc-D-Chg-OH Visit Supplier Webpage Request for quotationCAS: 198543-96-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: FMOC-D-CHG-OH Request for quotation
CAS: 198543-96-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 198543-96-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: FMOC-D-CHG-OH Visit Supplier Webpage Request for quotation
CAS: 198543-96-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 198543-96-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


