191114-48-4
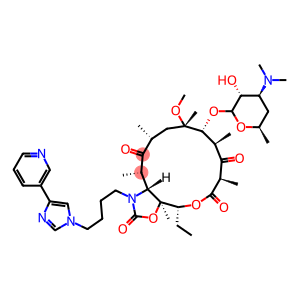
Telithromycin
CAS: 191114-48-4
Molecular Formula: C43H65N5O10
191114-48-4 - Names and Identifiers
191114-48-4 - Physico-chemical Properties
| Molecular Formula | C43H65N5O10 |
| Molar Mass | 812 |
| Density | 1.26±0.1 g/cm3(Predicted) |
| Melting Point | 176-188 C |
| Boling Point | 966℃ |
| Flash Point | >110°(230°F) |
| Water Solubility | Sparingly soluble in water |
| Solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | Solid |
| Color | White to Light Beige |
| pKa | 10.85±0.70(Predicted) |
| Storage Condition | Sealed in dry,Store in freezer, under -20°C |
| Use | This product is for scientific research only and shall not be used for other purposes. |
191114-48-4 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| RTECS | KF4674500 |
| Hazard Class | 9 |
191114-48-4 - Reference Information
| macrolide antibiotics | telithromycin is a new macrolide antibiotic developed by people in recent years. it is the first antibacterial drug in the semi-synthetic macrolide-lincolamide-streptomycin B(MLSB) family and the first ketolactone antibiotic approved for clinical application. it was successfully developed by the French Sanofi-Avent Group, it has broad-spectrum antibacterial activity and low selective drug resistance. It has good curative effect on Streptococcus pneumoniae and its penicillin-resistant and erythromycin-resistant strains, pneumococcus, Haemophilus influenzae and Morahan. The antibacterial effect is better than that of azithromycin and other macrolide antibiotics. It is mainly used to treat respiratory infections, bronchitis, pharyngitis, tonsillitis and pneumonia. the mechanism of action of telithromycin is similar to that of macrolide antibiotics. it mainly inhibits protein synthesis and its translation and assembly by directly binding to the 50s subunit of bacterial ribosome. it can also bind to nucleotides in the ii and v structural regions of 23s ribosomal RNA. however, the biggest difference is that telithromycin has a binding force of about 10 times and 6 times stronger to wild ribosome than erythromycin and clarithromycin, respectively. The small difference in the modification of the V region of the ribosome structure increases the tolerance of MLSB antibiotics to bacteria by 20 times, thus making them effective against all resistant strains of macrolides. oral absorption is good, oral bioavailability is about 57%, food does not affect its absorption, 70% is metabolized by CYP3A4 in the liver to teritol, teritaric acid, N-deoxyamide derivatives, N-oxopyridine derivatives. T1/2 was 9.81h and renal clearance was 12.5L/h. There are many ways of excretion: 13% is excreted from urine in its original form, 3% is excreted from feces in its original form, and metabolites are excreted from liver in 37%. In patients with liver dysfunction, Cmax is reduced by about 20%,T1/2 is 1.4 times longer than that of normal people, and metabolic rate is also reduced. The average pharmacokinetic parameters measured in patients with community acquired pneumonia (CAP) are Cmax 2.89 mg/L,Cmin 0.19 mg/L and AUC 13.9 mg/(L h). On February 12, 2007, the U.S. Food and Drug Administration announced that it had made certain changes to the drug label of the antibiotic telithromycin (also known as Kenlik) produced by Sanofi-Avent, and the indications for this drug The two items of acute bacterial sinusitis and acute bacterial deterioration of chronic bronchitis will be removed from the drug instructions, and patients will be reminded to use telithromycin safely. |
| biological activity | Telithromycin(HMR3647) is an antibiotic that can act on community-acquired pneumonia. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 15:16:48
Supplier List
Product Name: Telithromycin Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Telithromycin Visit Supplier Webpage Request for quotationCAS: 191114-48-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Telithromycin Request for quotation
CAS: 191114-48-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 191114-48-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Telithromycin Request for quotation
CAS: 191114-48-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 191114-48-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: Telithromycin Visit Supplier Webpage Request for quotationCAS: 191114-48-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Telithromycin Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Telithromycin Visit Supplier Webpage Request for quotationCAS: 191114-48-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Telithromycin Request for quotation
CAS: 191114-48-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 191114-48-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Telithromycin Request for quotation
CAS: 191114-48-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 191114-48-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: Telithromycin Visit Supplier Webpage Request for quotationCAS: 191114-48-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




