186381-39-5
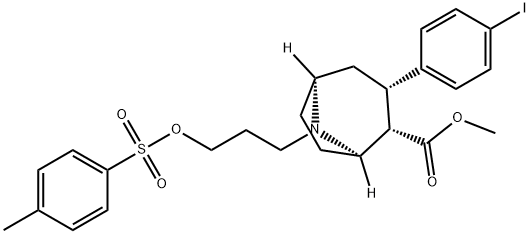
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy]propyl]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1)
CAS: 186381-39-5
Molecular Formula: C25H30INO5S
186381-39-5 - Names and Identifiers
| Name | 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy]propyl]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1) |
| Synonyms | TPCI TPCI186381-39-5 |
| CAS | 186381-39-5 |
186381-39-5 - Physico-chemical Properties
| Molecular Formula | C25H30INO5S |
| Molar Mass | 583.48 |
186381-39-5 - Introduction
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylsulfonfonic acid (1:1) is a 4-triphenylpterosanthene, also known as Triphenylchloromethane or Tritylchloride, with the chemical formula (C6H5)3CCL. The following is about 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1) introduction of the nature, use, preparation and safety information:
Nature:
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1) is a white crystal or powder, odorless, and has strong irritation. It has a low solubility, almost insoluble in water at room temperature, but soluble in organic solvents such as ether, chlorinated hydrocarbons.
Use:
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzensulfonic acid (1:1) is often used as a reagent and intermediate in organic synthesis. It can be used to mediate acid-catalyzed reactions, and can be used as a deprotection reagent for the removal of protective groups of ether, Ester and alcohol compounds. In addition, 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzensulfonic acid (1:1) can also be used in other occasions of organic synthesis, such as the synthesis of aromatic compounds and organometallic chemical reactions.
Method:
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1) can be prepared by reacting triphenylmethanol with thionyl chloride. During the reaction, thionyl chloride is used as the chlorine source for the reaction, and triphenylmethanol is chlorinated to form 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)) sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzensulfonic acid (1:1). Specific reaction conditions and operating methods can be referred to the professional organic synthesis literature.
Safety Information:
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzensulfonic acid (1:1) is irritating and should avoid contact with skin and eyes. Wear protective gloves and goggles during operation. In addition, 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1) is also toxic and should be kept away from food, beverages and organic matter. When storing and handling 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1), appropriate protective measures should be taken to avoid contact with oxidants and acidic substances, avoid producing toxic gases.
Nature:
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1) is a white crystal or powder, odorless, and has strong irritation. It has a low solubility, almost insoluble in water at room temperature, but soluble in organic solvents such as ether, chlorinated hydrocarbons.
Use:
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzensulfonic acid (1:1) is often used as a reagent and intermediate in organic synthesis. It can be used to mediate acid-catalyzed reactions, and can be used as a deprotection reagent for the removal of protective groups of ether, Ester and alcohol compounds. In addition, 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzensulfonic acid (1:1) can also be used in other occasions of organic synthesis, such as the synthesis of aromatic compounds and organometallic chemical reactions.
Method:
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1) can be prepared by reacting triphenylmethanol with thionyl chloride. During the reaction, thionyl chloride is used as the chlorine source for the reaction, and triphenylmethanol is chlorinated to form 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)) sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzensulfonic acid (1:1). Specific reaction conditions and operating methods can be referred to the professional organic synthesis literature.
Safety Information:
8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzensulfonic acid (1:1) is irritating and should avoid contact with skin and eyes. Wear protective gloves and goggles during operation. In addition, 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1) is also toxic and should be kept away from food, beverages and organic matter. When storing and handling 8-Azabicyclo[3.2.1]octane-2-carboxylic acid, 3-(4-iodophenyl)-8-[3-[[(4-methylphenyl)sulfonyl]oxy] Proxy]-, methyl ester, (1R,2S,3S,5S)-, salt with 4-methylbenzenesulfonic acid (1:1), appropriate protective measures should be taken to avoid contact with oxidants and acidic substances, avoid producing toxic gases.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: TPCI Request for quotation
CAS: 186381-39-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 186381-39-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: TPCI Request for quotation
CAS: 186381-39-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 186381-39-5
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
View History
