179898-50-1
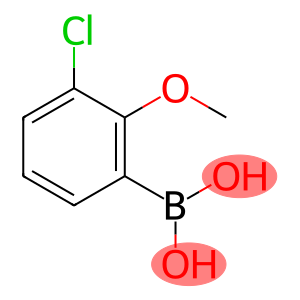
3-CHLORO-2-METHOXYPHENYLBORONIC ACID
CAS: 179898-50-1
Molecular Formula: C7H8BClO3
179898-50-1 - Names and Identifiers
179898-50-1 - Physico-chemical Properties
| Molecular Formula | C7H8BClO3 |
| Molar Mass | 186.4 |
| Density | 1.32±0.1 g/cm3(Predicted) |
| Boling Point | 356.9±52.0 °C(Predicted) |
| pKa | 7.58±0.58(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
179898-50-1 - Introduction
ACID is an organic boron compound with the chemical formula C7H7BClO2. It has the following properties:
1. Appearance: colorless or light yellow crystal;
2. Solubility: soluble in organic solvents such as methanol, ethanol and dichloromethane, slightly soluble in water;
3. Melting Point: about 135-140 ℃.
Ti ACID can be applied to boration reaction in organic synthesis reaction. It can be used as a Boronic ester reagent of aromatic compounds to construct the formation of carbon-boron bonds, thereby synthesizing various organic compounds. In addition, it can also be used as a fluorescent probe, a photosensitive dye and a synthetic intermediate of coordination compounds.
Preparation Cr ACID can be obtained by reacting 2-methoxybenzeneboronic ACID with boron trichloride. The reaction is generally carried out under dry and inert atmosphere.
1. Appearance: colorless or light yellow crystal;
2. Solubility: soluble in organic solvents such as methanol, ethanol and dichloromethane, slightly soluble in water;
3. Melting Point: about 135-140 ℃.
Ti ACID can be applied to boration reaction in organic synthesis reaction. It can be used as a Boronic ester reagent of aromatic compounds to construct the formation of carbon-boron bonds, thereby synthesizing various organic compounds. In addition, it can also be used as a fluorescent probe, a photosensitive dye and a synthetic intermediate of coordination compounds.
Preparation Cr ACID can be obtained by reacting 2-methoxybenzeneboronic ACID with boron trichloride. The reaction is generally carried out under dry and inert atmosphere.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: (3-Chloro-2-methoxyphenyl)boronic acid Visit Supplier Webpage Request for quotationCAS: 179898-50-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-CHLORO-2-METHOXYPHENYLBORONIC ACID Request for quotation
CAS: 179898-50-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 179898-50-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 3-CHLORO-2-METHOXYPHENYLBORONIC ACID Visit Supplier Webpage Request for quotation
CAS: 179898-50-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 179898-50-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (3-Chloro-2-methoxyphenyl)boronic acid Visit Supplier Webpage Request for quotationCAS: 179898-50-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-CHLORO-2-METHOXYPHENYLBORONIC ACID Request for quotation
CAS: 179898-50-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 179898-50-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 3-CHLORO-2-METHOXYPHENYLBORONIC ACID Visit Supplier Webpage Request for quotation
CAS: 179898-50-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 179898-50-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


