1744-22-5
riluzole
CAS: 1744-22-5
Molecular Formula: C8H5F3N2OS
1744-22-5 - Names and Identifiers
| Name | riluzole |
| Synonyms | riluzole 6-trifluoromethoxy-2-aminobenzothiazole 6-(TRIFLUOROMETHOXY)-2-BENZOTHIAZOLAMINE 2-Amino-6-(trifluoromethoxy)benzothiazole 6-TRIFLUOROMETHOXY-BENZOTHIAZOL-2-YLAMINE 2-AMINO-6-(TRIFLUOROMETHOXY)BENZOTHIAZOLE 6-TRIFLUOROMETHOXYBENZOTHIAZOLE-2-YL-AMINE 6-(TRIFLUOROMETHOXY)-1,3-BENZOTHIAZOL-2-AMINE 6-(trifluoromethoxy)-1,3-benzothiazol-2-amine 1-butyl-3-methyl-1H-imidazol-3-ium hexafluorophosphate 6-(trifluoromethoxy)-1,3-benzothiazol-2-amine hydrochloride |
| CAS | 1744-22-5 |
| EINECS | 605-724-6 |
| InChI | InChI=1/C8H5F3N2OS.ClH/c9-8(10,11)14-4-1-2-5-6(3-4)15-7(12)13-5;/h1-3H,(H2,12,13);1H |
| InChIKey | FTALBRSUTCGOEG-UHFFFAOYSA-N |
1744-22-5 - Physico-chemical Properties
| Molecular Formula | C8H5F3N2OS |
| Molar Mass | 234.2 |
| Density | 1.572±0.06 g/cm3(Predicted) |
| Melting Point | 116-118°C |
| Boling Point | 296.3±50.0 °C(Predicted) |
| Flash Point | 133°C |
| Solubility | DMSO: ≥25mg/mL |
| Vapor Presure | 0.00145mmHg at 25°C |
| Appearance | Powder |
| Color | white |
| Merck | 14,8223 |
| pKa | 2.96±0.10(Predicted) |
| Storage Condition | 2-8°C |
| Stability | Stable for 1 year from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 2 months. |
| Physical and Chemical Properties | Crystallization from ethanol-water (1:1), melting point 119 °c. Acute toxicity LD50 mice (mg/kg):46 intraperitoneal injection; 67 oral. |
1744-22-5 - Risk and Safety
| Risk Codes | 25 - Toxic if swallowed |
| Safety Description | 45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 2811 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | DL2830000 |
| HS Code | 29342000 |
| Hazard Note | Irritant |
| Hazard Class | 6.1 |
| Packing Group | II |
| Toxicity | LD50 in mice (mg/kg): 46 i.p.; 67 orally (Mizoule) |
1744-22-5 - Standard
Authoritative Data Verified Data
This product is 2-amino-6-trifluoromethoxybenzothiazole. The content of C8H5F3N2OS shall not be less than 98.5% calculated as anhydrous.
1744-22-5 - Trait
Authoritative Data Verified Data
- This product is white to yellowish crystal or crystalline powder; Odorless.
- This product is soluble in methanol or ethanol, and almost insoluble in water.
melting point
The melting point of this product (General 0612) is 117~119.5°C.
1744-22-5 - Differential diagnosis
Authoritative Data Verified Data
- take an appropriate amount of this product, add 0.lmol/L hydrochloric acid solution was dissolved and diluted to prepare a solution containing about l0ug per lml, which was determined by UV-Vis spectrophotometry (General rule 0401) at 254nm, there is a maximum absorption at the wavelength of 280nm and 287nm.
- The infrared absorption spectrum of this product should be consistent with that of the reference product (General rule 0402).
1744-22-5 - Exam
Authoritative Data Verified Data
clarity and color of ethanol solution
take an appropriate amount of this product, add ethanol to dissolve and dilute to make a solution containing about 10 mg per lml, the solution should be clear and colorless; If the color is developed, comparison with yellow No. 2 Standard Colorimetric solution (General rule 0901 first method), not deeper.
Related substances
take this product, add mobile phase to dissolve and dilute to make a solution containing about 0.5mg per lml as a test solution; Take lml for precision measurement and put it in a 100ml measuring flask, dilute to the scale with the mobile phase, shake, and serve as a control solution. According to the high performance liquid chromatography (General 0512) test, using eighteen alkyl silane bonded silica gel as filler, methanol-water (70:30) as mobile phase, the detection wavelength was 221mn, the number of theoretical plates is not less than 2000 based on riluzole peak. The LOLs of the test solution and the control solution were respectively injected into the human liquid chromatograph, and the chromatogram was recorded to 3 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.5 times (0.5%) the area of the main peak of the control solution, and the sum of the areas of each impurity peak shall not be greater than the area of the main peak of the control solution (1.0%).
moisture
take this product, according to the moisture determination method (General 0832 first method 1) determination, not over 0.5%.
ignition residue
take l.Og of this product and check it according to law (General rule 0841). The residue left shall not exceed 0.1%.
Heavy metals
The residue left under the item of taking the ignition residue shall not contain more than 10 parts per million of heavy metal when examined by law (General Principles 0821, Law II).
1744-22-5 - Content determination
Authoritative Data Verified Data
take this product about 0.19g, precision weighing, add glacial acetic acid 25ml and acetic anhydride 5ml to dissolve, add crystal violet indicator solution 1 drop, with perchloric acid titration solution (O. 1 mol/L) rapid titration to the solution is blue-green, and the titration results are corrected with a blank test. Per 1 ml perchloric acid titrant (0.1 mol/L) corresponds to 23.42mg of C8H5F3N2OS.
1744-22-5 - Category
Authoritative Data Verified Data
nervous system medication.
1744-22-5 - Storage
Authoritative Data Verified Data
sealed storage.
1744-22-5 - Riluzole Tablets
Authoritative Data Verified Data
This product contains riluzole (C8H5F3N2OS) should be labeled the amount of 95.0% to 105.0%.
trait
This product is a white or white-like tablet or film-coated tablet, which shows a white color after removing the coating.
identification
- a solution containing about 10ug of riluzole per 1 ml was prepared by diluting the sample solution under the item of related substances with the mobile phase, the mobile phase was added to dissolve and dilute to make a solution containing about 10ug per 1 ml. The retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution according to the chromatographic conditions under the relevant substances.
- the sample solution under the content measurement item was taken and measured by ultraviolet-visible spectrophotometry (General 0401), and the maximum absorption was found at wavelengths of 254nm, 280nm and 287nm.
- This product shows the identification reaction of organic fluoride (General rule 0301).
examination
- relevant substances: take an appropriate amount of fine powder of this product, add mobile phase to dissolve riluzole and dilute it to make a solution containing 0.5mg riluzole per 1ml, filter it, and take the continued filtrate as the test solution; take 1ml of precision, 100ml flask, diluted with mobile phase to scale, shake, as a control solution. The determination was carried out according to the method for riluzole related substances. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.5 times (0.5%) the area of the main peak of the control solution, and the sum of the areas of each impurity peak shall not be greater than the area of the main peak of the control solution (1.0%).
- dissolution of this product, according to the dissolution and release determination method (General 0931 second method), with 0.900ml of 1 mol/L hydrochloric acid solution is the dissolution medium, and the rotation speed is 50 revolutions per minute. The operation is carried out according to law. After 30 minutes, the appropriate amount of the solution is taken, filtered, and 5ml of the continuous filtrate is taken in a precise amount, in a 25ml measuring flask, use 0. Dilute the lmol/L hydrochloric acid solution to the scale, shake well, and measure the absorbance at the wavelength of 254nm by UV-Vis spectrophotometry (General rule 0401), precision weighing, add 0.1 mol/L hydrochloric acid solution was dissolved and quantitatively diluted to prepare a solution containing about Loug per 1 ml, which was measured by the same method, and the dissolution amount of each tablet was calculated. The limit is 80% of the labeled amount and shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
Take 20 tablets of this product, precision weighing, fine grinding, precision weighing fine powder appropriate amount (about 50mg equivalent to riluzole), put in 250ml measuring flask, add 0.1 mol/L hydrochloric acid solution to dissolve riluzole (if necessary, ultrasonic) and dilute to the scale, shake, filter, Take 5ml of continuous filtrate, put it in a 100ml measuring flask, use 0. Dilute 1 mol/L hydrochloric acid solution to the scale, shake well, as the test solution, according to UV-visible spectrophotometry (General 0401), at the wavelength of 254mn absorbance; another riluzole control, precision weighing, plus O.lmol/L hydrochloric acid solution is dissolved and quantitatively diluted to make a solution containing about 10ug per lml, which is determined and calculated by the same method.
category
with riluzole.
specification
50mg
storage
light shielding, sealed storage.
1744-22-5 - Reference Information
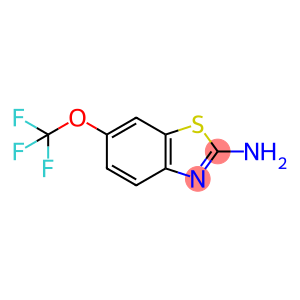
| introduction | riluzole is a benzothiazole compound and is currently the only drug approved by the U.S. Food and Drug Administration (FDA) for the treatment of amyotrophic lateral sclerosis. It mainly works by regulating the concentration of glutamate and acting on ion channels. Riluzole has a wide range of pharmacological effects, such as regulating glutamate and its transporters, neuroprotective effects, antidepressant, anti-anxiety, analgesic, etc. Figure 1 is the structural formula of riluzole |
| pharmacological effects | riluzole has the following physiological functions: 1. regulation of glutamate riluzole is an important glutamate regulator, which affects the concentration of glutamate in the intercellular space from different links. It affects the concentration of glutamate by affecting the activity of glutamate transporters. It can weaken the activity of excitatory amino acid transporter 3, and has a linear relationship in the concentration range of 0.3~100 μmol · L-1 (the concentration given to oocytes), indicating that riluzole through protein kinase C(protein kinaseC,PKC) inhibition to weaken the activity of excitatory amino acid transporter 3. 2. neuroprotective effect in various animal models of neuronal injury mediated by excitatory neurotoxicity, riluzole shows injury repair function, and it can also promote the production of various nerve growth factors. In the spinal muscular atrophy model, it improves the defective neuroprotective behavior of the spinal muscular atrophy model by activating potassium ion channels with small conductance calcium. 3. the effect on nerve discharge riluzole reduces the repetitive discharge caused by continuous current stimulation at different concentrations, but has little effect on the discharge caused by transient current. riluzole can activate TRPC5 heterologous expression in HEK293 cells and U-87 endogenous expression in malignant glioma cell lines. Although riluzole can activate other ion channels, it does not activate other TRPC members. Thus, riluzole is a useful pharmacological tool for the specialized recognition of TRPC5 currents in infinitely proliferated and acutely isolated primary cells. 4. Antidepressant Studies have found that riluzole combined with antidepressants can significantly improve the condition of patients with refractory depression. The linear mixed model shows that there is a certain relationship between the treatment time and the treatment effect. After 1 week of treatment, the effect is significant and can last for 12 weeks. In the treatment of bipolar depression, riluzole combined with lithium has achieved significant results, and the treatment process did not cause mania. 5. anti-anxiety numerous evidences confirm the important position of glutamatergic neurotransmitter system in the treatment of emotional and anxiety-like diseases. riluzole can regulate the concentration of glutamate in different links, so it is used for anti-anxiety research. To study the relationship between hippocampal volume and hippocampal N-acetylaspartic acid in the treatment of generalized anxiety disorder with riluzole. Preliminary research results show that hippocampal N-acetylaspartic acid and hippocampal volume are neurobiomarkers related to glutamate treatment response. 6. antiepileptic diphenylhydantoin, riluzole, lidocaine and other three Na channel blockers reduce hippocampal epileptiform activity through different mechanisms of action. The three compounds acted on different voltage-gated sodium channel sites, but produced similar reticular dominant excitability reductions. Diphenylhydantoin and riluzole continue to act on changes in network activity until the drug disappears completely. However, lidocaine can only increase the hippocampal CA3 cluster interval (inter-burst intervals,IBIs) at higher concentrations. Therefore, their function is not permanent to cause the hippocampal CA3 outbreak to return to zero. These data reveal that the hippocampal CA3 outbreak provides a relatively simple and fast platform for identifying compounds that can reduce network excitability and provide preliminary screening for further more complex in vivo animal studies. 7. analgesic effect dorsal root ganglion (DRG), as the first level neuron of pain afferent, plays an important role in the peripheral mechanism of pain. Studies have shown that riluzole can promote the growth of DRG neurites. Figure 2 shows riluzole |
| pharmacokinetics | after oral administration, the product can be quickly absorbed and reach the peak blood concentration within 60~90min after administration. About 90% of the drug is absorbed with an absolute bioavailability of 60%. This product is metabolized by liver and mainly excreted in urine. The elimination half-life is 9~15h. (2015-11-17) |
| indication | is suitable for the treatment of amyotrophic lateral sclerosis, prolonging life or mechanical ventilation time. But not for other forms of motor neuron disease. |
| side effects | taking riluzole has the following side effects: 1. common fatigue, stomach discomfort and elevated plasma transaminase level. 2. Stomach pain, headache, vomiting, increased heartbeat, dizziness, drowsiness, allergic reactions or pancreatitis (pancreatitis) are rare. 3. Occasionally neutropenia. |
| taboo | 1. those who are allergic to this product are prohibited. 2. The transaminase level is 3 times or more higher than the normal value is prohibited. 3. Pregnant and lactating women are prohibited. |
| precautions | 1. patients with renal insufficiency, neutropenia and hypertension should be used with caution. 2. Medication should be taken 1 hour before or 2 hours after meals to reduce the effect of food on the bioavailability of riluzole. 3. Liver function should be checked regularly during medication. 4. Dizziness or dizziness may occur after taking. You should not drive a vehicle or operate a machine. 5. There is no research data on children's application of this product, so it is not recommended. 6. Increasing the daily dose will not increase the efficacy, but increase the adverse reactions. If you miss it once, take the next dose as originally planned. |
| drug interaction | 1.CYP1A2 inhibitors (caffeine, phenacetin, theophylline, amitriptyline and quinolones) may reduce the clearance of this drug. 2.CYP1A2 inducers (rifampicin, omeprazole) may increase the clearance of this drug. |
| preparation specification | tablet, 50mg. |
| use | glutamate antagonist is a therapeutic drug for motor nerve diseases. For the treatment of amyotrophic lateral sclerosis (ALS). |
| Production method | 4-trifluoromethoxyaniline and NH4CNS are reacted in acetic acid, and then reacted with bromine at 5~10 ℃ overnight to obtain the product, the yield is 75%, and the melting point is 117~119 ℃. |
Supplier List
CAS: 1744-22-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 1744-22-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 1744-22-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1744-22-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1744-22-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 1744-22-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 1744-22-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1744-22-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025



