15861-40-2
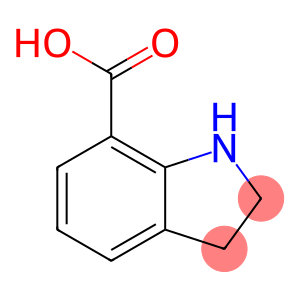
1H-Indole-7-carboxylic acid, 2,3-dihydro-
CAS: 15861-40-2
Molecular Formula: C9H9NO2
15861-40-2 - Names and Identifiers
15861-40-2 - Physico-chemical Properties
| Molecular Formula | C9H9NO2 |
| Molar Mass | 163.17 |
| Density | 1.283±0.06 g/cm3(Predicted) |
| Melting Point | 156 °C (decomp) |
| Boling Point | 356.3±31.0 °C(Predicted) |
| pKa | 1.87±0.20(Predicted) |
15861-40-2 - Introduction
1H-Indole-7-carboxylic acid, 2,3-dihydro-is an organic compound with the chemical formula C9H6N2O2. The following is a description of its nature, use, preparation and safety information:
Nature:
1. Appearance: 1H-Indole-7-carboxylic acid, 2,3-dihydro-is a white to yellow crystalline powder.
2. Melting Point: about 221-223 ℃.
3. Solubility: It dissolves in most common organic solvents, such as dimethyl sulfoxide, ethanol and dichloromethane. But its solubility in water is low.
Use:
1H-Indole-7-carboxylic acid, 2,3-dihydro-has a variety of applications in the chemical and pharmaceutical fields:
1. As an intermediate in Organic Synthesis: It can be used as an important intermediate for the synthesis of a variety of drugs and bioactive molecules.
2. Biochemical research: Because 1H-Indole-7-carboxylic acid, 2,3-dihydro-has strong fluorescence properties, it can be used for staining or biomarkers in vivo.
3. Anti-tumor drug research: Some studies have shown that 1H-Indole-7-carboxylic acid, 2,3-dihydro-may have anti-tumor activity, so it has certain potential in anti-cancer drug research.
Method:
The main method for preparing 1H-Indole-7-carboxylic acid, 2,3-dihydro-is obtained by reacting indoline with carbon dioxide under high pressure. This reaction generally requires high temperatures and the presence of a catalyst.
Safety Information:
1. 1H-Indole-7-carboxylic acid, 2,3-dihydro-irritating to skin and eyes, should pay attention to protection when touching. In the process of operation, should follow good laboratory safety procedures, including wearing appropriate protective equipment (such as gloves, goggles).
3. Since 1H-Indole-7-carboxylic acid, 2,3-dihydro-is an organic synthesis intermediate in the laboratory, it is necessary to follow appropriate storage and disposal methods to ensure safety and environmental protection.
Nature:
1. Appearance: 1H-Indole-7-carboxylic acid, 2,3-dihydro-is a white to yellow crystalline powder.
2. Melting Point: about 221-223 ℃.
3. Solubility: It dissolves in most common organic solvents, such as dimethyl sulfoxide, ethanol and dichloromethane. But its solubility in water is low.
Use:
1H-Indole-7-carboxylic acid, 2,3-dihydro-has a variety of applications in the chemical and pharmaceutical fields:
1. As an intermediate in Organic Synthesis: It can be used as an important intermediate for the synthesis of a variety of drugs and bioactive molecules.
2. Biochemical research: Because 1H-Indole-7-carboxylic acid, 2,3-dihydro-has strong fluorescence properties, it can be used for staining or biomarkers in vivo.
3. Anti-tumor drug research: Some studies have shown that 1H-Indole-7-carboxylic acid, 2,3-dihydro-may have anti-tumor activity, so it has certain potential in anti-cancer drug research.
Method:
The main method for preparing 1H-Indole-7-carboxylic acid, 2,3-dihydro-is obtained by reacting indoline with carbon dioxide under high pressure. This reaction generally requires high temperatures and the presence of a catalyst.
Safety Information:
1. 1H-Indole-7-carboxylic acid, 2,3-dihydro-irritating to skin and eyes, should pay attention to protection when touching. In the process of operation, should follow good laboratory safety procedures, including wearing appropriate protective equipment (such as gloves, goggles).
3. Since 1H-Indole-7-carboxylic acid, 2,3-dihydro-is an organic synthesis intermediate in the laboratory, it is necessary to follow appropriate storage and disposal methods to ensure safety and environmental protection.
Last Update:2024-04-09 21:04:16
Supplier List
Product Name: 1H-INDOLE-7-CARBOXYLIC ACID,2,3-DIHYDRO- Request for quotation
CAS: 15861-40-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 15861-40-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 1H-INDOLE-7-CARBOXYLIC ACID,2,3-DIHYDRO- Visit Supplier Webpage Request for quotation
CAS: 15861-40-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 15861-40-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 1H-INDOLE-7-CARBOXYLIC ACID,2,3-DIHYDRO- Request for quotation
CAS: 15861-40-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 15861-40-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 1H-INDOLE-7-CARBOXYLIC ACID,2,3-DIHYDRO- Visit Supplier Webpage Request for quotation
CAS: 15861-40-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 15861-40-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

