151-10-0
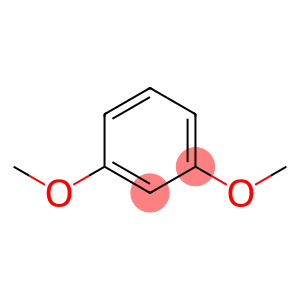
1,3-Dimethoxy benzene
CAS: 151-10-0
Molecular Formula: C8H10O2
151-10-0 - Names and Identifiers
151-10-0 - Physico-chemical Properties
| Molecular Formula | C8H10O2 |
| Molar Mass | 138.16 |
| Density | 1.055g/mLat 25°C(lit.) |
| Melting Point | -52°C |
| Boling Point | 85-87°C7mm Hg(lit.) |
| Flash Point | 190°F |
| JECFA Number | 1249 |
| Water Solubility | 1.216g/L(25 ºC) |
| Solubility | Miscible with toluene. |
| Vapor Presure | 0.195mmHg at 25°C |
| Appearance | Liquid |
| Color | Clear colorless to slightly brown |
| BRN | 878582 |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | n20/D 1.524(lit.) |
| Physical and Chemical Properties | Density 1.055 boiling point 85-87°C (7 torr) refractive index 1.523-1.525 flash point 87°C |
| Use | It is used as an intermediate for naphthalene dyes, paints and plastics, and as a fragrance for soaps, detergents and ointments. |
151-10-0 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| UN IDs | NA 1993 / PGIII |
| WGK Germany | 2 |
| RTECS | CZ6474000 |
| TSCA | Yes |
| HS Code | 29093090 |
| Hazard Note | Irritant |
151-10-0 - Upstream Downstream Industry
| Raw Materials | Dimethyl sulfate |
151-10-0 - Reference Information
| FEMA | 2385 | M-DIMETHOXYBENZENE |
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Use | 1, 3-dimethoxybenzene is an intermediate for the synthesis of the acaricide benzofurate, and can also be used for the preparation of UV absorber UV-9. It is used as an intermediate for naphthalene dyes and coatings and plastics. It is also used as a fragrance for soaps, detergents and ointments. used as a pharmaceutical intermediate used as an intermediate in organic synthesis, a f-g reaction was carried out with benzoyl chloride under the catalysis of aluminum trichloride to obtain 2, 4-dimethoxybenzophenone. This is an intermediate of ultraviolet absorber UV-9. |
| production method | is obtained by reacting resorcinol with dimethyl sulfate. 1, 3-dimethoxybenzene is prepared by the reaction of resorcinol with dimethyl sulfate. Resorcinol is mixed with sodium hydroxide solution, dimethyl sulfate is added under stirring, methylation reaction is carried out at 26~30 ° C., and then separation, washing and distillation are carried out to obtain the finished product. In the laboratory preparation, resorcinol (1mol), 200ml of methanol and dimethyl sulfate (1mol) were added to the reactor, stirred and dissolved, cooled to -5 °c with ice salt bath, A solution of 150g of potassium hydroxide and 350ml of water was immediately added, and a vigorous reaction occurred, and the methylation reaction was completed within 3min. After stirring for 5min, water was added to dilute, and the oil layer was separated. The water layer was extracted with ether, the oil layer and ether extract were combined, washed with water, dried with anhydrous calcium chloride, and the ether was recovered on a water bath, then it was heated and distilled by direct heat, and the fraction at 216~218 °c was collected, and 124g of 1, 3-dimethoxybenzene was obtained, with a yield of 90%. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 1,3-Dimethoxybenzene Visit Supplier Webpage Request for quotationCAS: 151-10-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 1,3-Dimethoxybenzene Request for quotation
CAS: 151-10-0
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 151-10-0
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Spot supply
Product Name: m-Dimethoxy benzene Visit Supplier Webpage Request for quotationCAS: 151-10-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: Dimethoxybenzene Request for quotationCAS: 151-10-0
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Product Name: Dimethoxybenzene Request for quotation
CAS: 151-10-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 151-10-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 1,3-Dimethoxy benzene Request for quotation
CAS: 151-10-0
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 151-10-0
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: 1,3-Dimethoxybenzene Visit Supplier Webpage Request for quotationCAS: 151-10-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 1,3-Dimethoxybenzene Request for quotation
CAS: 151-10-0
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 151-10-0
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Spot supply
Product Name: m-Dimethoxy benzene Visit Supplier Webpage Request for quotationCAS: 151-10-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: Dimethoxybenzene Request for quotationCAS: 151-10-0
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Product Name: Dimethoxybenzene Request for quotation
CAS: 151-10-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 151-10-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 1,3-Dimethoxy benzene Request for quotation
CAS: 151-10-0
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 151-10-0
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
View History
Raw Materials for 151-10-0





