149908-53-2
Azimilide
CAS: 149908-53-2
Molecular Formula: C23H28ClN5O3
149908-53-2 - Names and Identifiers
| Name | Azimilide |
| Synonyms | NE-10064 AZIMILIDE Azimilide UNII-74QU6P2934 Unii-74qu6p2934 Azimilide(NE-10064) 2,4-Imidazolidinedione, 1-(((5-(4-chlorophenyl)-2-furanyl)methylene)amino)-3-(4-(4-methyl-1-piperazinyl)butyl)- |
| CAS | 149908-53-2 |
| InChI | InChI=1/C23H28ClN5O3/c1-26-12-14-27(15-13-26)10-2-3-11-28-22(30)17-29(23(28)31)25-16-20-8-9-21(32-20)18-4-6-19(24)7-5-18/h4-9,16H,2-3,10-15,17H2,1H3/b25-16+ |
149908-53-2 - Physico-chemical Properties
| Molecular Formula | C23H28ClN5O3 |
| Molar Mass | 457.95 |
| Density | 1.32 |
| Boling Point | 594.9±60.0 °C(Predicted) |
| pKa | 7.72±0.10(Predicted) |
| Storage Condition | 2-8°C |
| Physical and Chemical Properties | Azmil Dihydrochloride: C23H28C1N5O3? 2HCl. [149888-94-8]. Crystallization from ethanol-water. |
| Use | Antiarrhythmic drugs |
149908-53-2 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 1.884 ml | 9.418 ml | 18.837 ml |
| 5 mM | 0.377 ml | 1.884 ml | 3.767 ml |
| 10 mM | 0.188 ml | 0.942 ml | 1.884 ml |
| 5 mM | 0.038 ml | 0.188 ml | 0.377 ml |
Last Update:2024-01-02 23:10:35
149908-53-2 - Reference Information
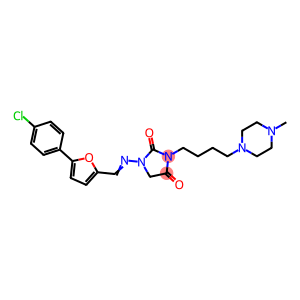
| antiarrhythmic drugs | atrial fibrillation is the most common arrhythmia. patients not only reduce cardiac output, but also increase the risk of stroke death by 2 and 5 times respectively. A large number of clinical observations show that timely conversion of sinus rhythm and effective maintenance is the best treatment option. There are two ways to convert sinus rhythm, electrical and drug, and only one drug treatment is to maintain sinus rhythm. Therefore, the development of new drugs for the treatment of atrial fibrillation has its urgency and importance. Class l A antiarrhythmic drugs (quinidine, procainamide, propiamide) were once the only drugs to convert and maintain sinus rhythm, but serious adverse reactions obviously restricted its wide clinical application. Studies have shown that the mortality rate of long-term quinidine is significantly higher than that of the placebo group. Since then, class lC (propafenone, flecainide) and class III antiarrhythmic drugs (amiodarone) have been clinically regarded as the first choice for long-term arrhythmia prevention due to their good tolerable efficacy and safety. However, IC antiarrhythmic drugs still have adverse reactions of negative inotropic and conduction inhibition, while amiodarone has a high incidence of systemic adverse reactions although the above-mentioned adverse reactions are less. Recent studies have shown that the shortening of atrial refractory period is the electrophysiological basis of atrial fibrillation. Therefore, the development and development of class III antiarrhythmic drugs is mainly aimed at the treatment of atrial fibrillation. The combined mechanism of class III antiarrhythmic drugs is to specifically block delayed rectifier potassium channel current (Ik), prolong myocardial action potential time course (APD) and effective refractory period (ERP), thus prolonging myocardial repolarization. The first-generation drugs represented by amiodarone and sotalol and the second-generation drugs represented by dofetilide and ibutilide are limited in clinical application due to long-term use, large adverse reactions or reverse frequency dependence (reverserate dependent). Reverse frequency dependence is manifested as: with the acceleration of heart rate, the effect of drugs to prolong APD and ERP is weakened or even disappeared, and its effect is enhanced when the heart rate slows down, which is not conducive to the full play of anti-tachyarrhythmia effect of drugs. On the contrary, it is easy to have arrhythmia effect when the heart rate slows down, resulting in prolonged QT interval and torsade de pointes (Tdp). The generation of reverse frequency dependence is believed to be caused by these drugs only specifically blocking the fast delayed rectifier potassium channel current, which causes the slow delayed rectifier potassium channel current to be strongly up-regulated. Azilide is a new class III antiarrhythmic drug, which can simultaneously block the fast-activating component (Ikr) and slow-activating component (Iks) in the delayed rectified outward potassium current, and prolong the action potential duration and effective refractory period. The larger SHIELD test found that 125mg azilite can reduce the risk of 55% electric storms, while 75g azilite can only reduce 53% electric storms caused by ICD discharge. However, neither dose of azilit could reduce the time of the first electric storm. Notably, tip torsion ventricular tachycardia (TdP) occurred in 2 cases (4%) in the 125mg group. This is related to the increase of Ca2 + influx in the plateau by Azilit, which is easy to cause calcium load and induce early and post depolarization (EAD). Fig. 1 is the structural formula of azilite |
| Pharmacological effects and mechanisms | 1. The effect on ion channels and the frequency-dependent chemical structure of azilide is different from other second-generation drugs that simply block Ikr, such as dofelide and ibutilide, which lack the mesulfonamide structure and are chlorobenzene furan compounds, it is the first artificially developed drug that can simultaneously block Ikr and Iks, and prolong the duration of myocardial action potential and effective refractory period. Azilit can block Ikr and Iks of canine ventricular myocytes at the concentration level of 10-6 mol.L-1, and the reverse frequency-dependent effect is low. Further research shows that the semi-inhibitory concentration (IC50) of azilite on Ikr is 0.1-0.4 μmol.L-1, and the IC50 on IkS is 0.7-3 μmol.L-1, and its blocking Iks effect is dose-dependent, time-dependent and very small reverse frequency-dependent. In a canine model of surgically induced myocardial infarction, iv azilite 10-30ng/kg, azilite can effectively increase the action potential duration and effective refractory period of canine ventricular myocytes at both faster and slower heart rates. In atrial arrhythmia, the effect of prolonging the action potential duration and effective refractory period has no obvious relationship with the speed of heart rate, while the effect of dofetilide is obviously weakened when the heart rate is faster. Studies have confirmed that azilide can block the Ikr and Iks components of the outward potassium flow in human atrial and ventricular myocytes, including in the case of sympathetic nerve excitation or nervous tension-induced tachycardia. At this time, the effect of class 1 drugs (such as ibutilide) that specifically block Ikr has been significantly weakened, and this product can still play a role. Among the antiarrhythmic effects of azilite, it is difficult to clearly distinguish which channel has a greater proportion of blocking effects. Azilit also has a blocking effect on L-type Ca2 + channels and inward sodium flow, and its IC50 is 10 times and 5 times the blocking Iks concentration, respectively. 2. Effects on receptors The study of radioactive sources in mice shows that azilite can also show the properties of β-blockers and α-receptors, toxinine receptors and 5-hydroxytryptamine (5-HT) receptor agonists. However, the β-blocker nature exhibited by azilide at conventional antiarrhythmic doses was not confirmed in anesthetized mouse experiments. |
| pharmacodynamic | delayed rectification of outward potassium ions in myocardial tissue is the main ion flow causing repolarization. this ion flow mainly includes Ikr and Iks. unlike other class III antiarrhythmic drugs, azilide also has a blocking effect on Iks, which works by non-selectively blocking Ik. Azilite can prolong myocardial repolarization, which is mainly manifested in the prolongation of action potential time course (APD) and effective refractory period (ERP). In addition, RR, QT and QTc intervals can be prolonged to different degrees, while PR interval and QRS complex have no obvious changes. 1. Oral administration: In a randomized controlled trial of 119 volunteers, the oral azilide load, bid,3 days later, 35, 100, 150, 200 mg and placebo, qd were given respectively. After 11 days of maintenance, it was found that its pharmacological effect was dose-dependent, and the maximum QTC interval was prolonged by 12.4%-14.6% when the dose was maintained at 10mg/d, without age and gender differences. Ten of all volunteers withdrew due to asymptomatic arrhythmia, including non-persistent ventricular tachycardia, atrial tachycardia, atrial flutter and atrial fibrillation. The incidence of arrhythmia was 8% in the test group and 9% in the control group. The safety and tolerability of oral azilite in more than 500 volunteers showed that after 12 hours of oral azilite 0.2-8 mg/kg, the QTc interval was prolonged 32% at the maximum dose of 8 mg/kg, and there was no obvious change in PR, QRS interval, heart rate and blood pressure, no serious arrhythmia and other adverse reactions, indicating good tolerance. In a randomized controlled trial, 384 patients with atrial flutter or atrial fibrillation who had converted to sinus rhythm were randomly divided into azilide 50, 100, 125 mg groups and placebo groups. The above dose of 3d(bid) was given before qd was maintained. After 4 days, it was found that RR, QT and QTC intervals in each test group were prolonged to varying degrees, and showed dose correlation. Among them, compared with placebo group, the average RR interval change values of 125mg group were +61.4 ms and-14.lms, the average QT interval change values were +44.2ms and -1.0ms, and the average QTC interval change values were +31.6 ms and + 2.l ms, all of which were significantly different (p 2. intravenous administration: azilite 0.6 mg/kg,iv, until the blood drug concentration reaches 54 mg/kg, it is observed that the QT interval is as long as the highest value 34% when the blood drug concentration is 8.9 mg/kg (p 4 μg/mL, the average maximum QTc interval is only extended by 24%-32%, indicating that the drug is safe and well tolerated. |
| use | is used to prevent recurrence of atrial flutter, atrial fibrillation and supraventricular tachycardia and to reduce the risk of sudden arrhythmic death in patients with recent myocardial infarction. (2016-06-17) Class III antiarrhythmic drugs. Potassium channel blockers. |
| adverse reactions | 1. in some cases, ventricular premature beats, ventricular tachycardia and Tdp can be seen. 2. The most common extracardiac adverse reactions are headache, followed by fatigue and dyspnea. |
| drug interaction | 1. after 18 patients treated with digoxin took azilide for 5 days to reach steady state, the absorption rate and renal clearance rate of digoxin were slightly increased, but there was no clinical significance and dose adjustment was not required. 2. When combined with warfarin, no obvious pharmacokinetic changes are found, and dose adjustment is not required. |
| Production method | Compound (I) reacts with l-bromo-4-chlorobutane, and the product (II) is converted into iodide with sodium iodide (III), and then reacts with 1-methylpiperazine to obtain compound (IV). Then it is hydrogenated and reduced to amine (V), and then combined with compound (VI) to form Schiff base to obtain the product. The product is obtained in the form of dihydrochloride. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: AZIMILIDE Request for quotation
CAS: 149908-53-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 149908-53-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: AZIMILIDE Visit Supplier Webpage Request for quotation
CAS: 149908-53-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 149908-53-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: AZIMILIDE Request for quotation
CAS: 149908-53-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 149908-53-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: AZIMILIDE Visit Supplier Webpage Request for quotation
CAS: 149908-53-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 149908-53-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

