145040-37-5
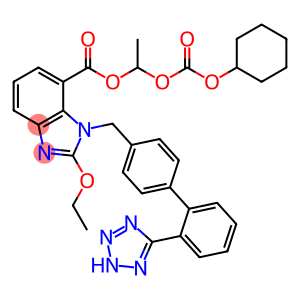
Candesartan cilexetil
CAS: 145040-37-5
Molecular Formula: C33H34N6O6
145040-37-5 - Names and Identifiers
| Name | Candesartan cilexetil |
| Synonyms | tcv116 Candesantan Cilexetil Candesartan cilexetil 1-(((cyclohexyloxy)carbonyl)oxy)ethylester,(+-)-henyl)-4-yl)methyl) 1h-benzimidazole-7-carboxylicacid,2-ethoxy-1-((2'-(1h-tetrazol-5-yl)(1,1'-bip 1-(Cyclohexyloxycarbonyloxy)ethyl 1-((2'-(1H-tetrazol-5-yl)biphenyl-4-yl)methyl)-2-ethoxy-1H-b 1-(((Cyclohexyloxy)carbonyl)oxy)ethyl 1-((2'-(2H-tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)-2 1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-ethoxy-1-[[2'-(1H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-1H-benzimidazole-7-carboxylate 1-(((cyclohexyloxy)carbonyl)oxy)ethyl 1-((2'-(1H-tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)-2-ethoxy-1H-benzo[d]imidazole-7-carboxylate 1H-Benzimidazole-7-carboxylic acid, 2-ethoxy-1-[[2'-(2H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-, 1-[[(cyclohexyloxy)carbonyl]oxy]ethyl ester |
| CAS | 145040-37-5 |
| EINECS | 627-030-2 |
| InChI | InChI=1/C33H34N6O6/c1-2-42-32-34-28-14-8-13-27(31(40)43-19-20-44-33(41)45-24-9-4-3-5-10-24)29(28)39(32)21-22-15-17-23(18-16-22)25-11-6-7-12-26(25)30-35-37-38-36-30/h6-8,11-18,24H,2-5,9-10,19-21H2,1H3,(H,35,36,37,38) |
| InChIKey | GHOSNRCGJFBJIB-UHFFFAOYSA-N |
145040-37-5 - Physico-chemical Properties
| Molecular Formula | C33H34N6O6 |
| Molar Mass | 610.66 |
| Density | 1.37±0.1 g/cm3(Predicted) |
| Melting Point | 168-170?C |
| Boling Point | 843.3±75.0 °C(Predicted) |
| Solubility | Soluble in water (<1 mg/ml at 25 °C), DMSO (122 mg/ml at 25 °C), methanol, DMF (~30 |
| Appearance | powder |
| Color | white to beige |
| Maximum wavelength(λmax) | ['304nm(EtOH)(lit.)'] |
| Merck | 14,1739 |
| pKa | pKa 3.55 (H2O t=25.0 I=0.025) (Uncertain);5.91(H2O t=25.0 I=0.025) (Uncertain) |
| Storage Condition | 2-8°C |
| Sensitive | Sensitive to heat |
| Refractive Index | 1.665 |
| MDL | MFCD00871371 |
| Use | Uses: pesticides and pharmaceutical intermediates |
145040-37-5 - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. R50 - Very Toxic to aquatic organisms R48/20 - R61 - May cause harm to the unborn child |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S53 - Avoid exposure - obtain special instructions before use. |
| WGK Germany | 3 |
| RTECS | DD6672500 |
| HS Code | 29339953 |
145040-37-5 - Standard
Authoritative Data Verified Data
This product is (±)-l-[(cyclohexyloxy) carbonyloxy] ethyl 2-ethoxy-1 [[2-1h-tetrazolyl-5-yl) biphenyl-4-yl] methyl]-1H-benzimidazole-7-carboxylic acid ester. Calculated as dry product, containing no less than 98.5% of C33H34N606.
145040-37-5 - Trait
Authoritative Data Verified Data
- This product is white or off-white crystalline powder.
- This product is soluble in chloroform, slightly soluble in anhydrous ethanol, and almost insoluble in water.
145040-37-5 - Differential diagnosis
Authoritative Data Verified Data
(1) take the control solution under the item of related substances as the test solution; Take the appropriate amount of Candesartan Cilexetil, add acetonitrile-water (3:2) A solution containing about 4ug per 1 ml was prepared as a control solution by dissolution and dilution. The retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the sample solution according to the chromatographic conditions of the related substances.
(2) the infrared absorption spectrum of this product should be consistent with the spectrum of the control (Spectrum set 1147 figure).
145040-37-5 - Exam
Authoritative Data Verified Data
chloride
take 0.40g of this product, add 0801 of water, strongly shake for 10 minutes, filter, take 25ml of filtrate, check according to law (general rule), and standard sodium chloride solution 5.0ml of the control solution should not be deeper (0.05%).
Related substances
take about 20mg of this product, put it in a 50ml measuring flask, add acetonitrile-water (3:2) to dissolve and dilute to the scale, shake well, as a test solution; 1 ml was accurately measured, placed in a 100ml measuring flask, diluted to the scale with acetonitrile-water (3:2), and shaken to serve as a control solution. Using octadecyl cinnamate-bonded cinnamate as filler (Ecosd C18 column or Kromasil100-5 C18 column, 0512 × 4.6) as determined by HPLC (general), 5pm or performance equivalent column); Acetonitrile-glacial acetic acid-water (57:1:43) as mobile phase A, acetonitrile-glacial acetic acid-water (90:1:10) mobile Phase B, the detection wavelength was 254nm. Gradient elution was performed as follows. Take 20ml of test solution and add 0.lmol/L hydrochloric acid solution l. 0ML, 90°C water bath heating for 10 minutes, add 0.1.0mL of 1 mol/L sodium hydroxide solution was neutralized and used as the system applicable solution, 10u1 was injected into the liquid chromatograph, and the chromatogram was recorded. The peak retention time of Candesartan Cilexetil was about 20 minutes, (±)-1 -[(cyclohexyloxy) carbonyloxy] ethyl 2-oxotetrazol-5-yl) biphenyl-4-yl] methyl]-2, the relative retention time of 3-dihydro-1h-benzimidazole-4-carboxylate (impurity I) is about 0.6; The number of theoretical plates is not less than 12 000 in terms of Candesartan Cilexetil peak, and the tailing factor should not exceed 1.5. Accurately take 10ul of each sample solution and control solution into the liquid chromatograph, record the chromatogram, if there are impurity peaks in the chromatogram of the sample solution, the Peak area of impurity I shall not be greater than 0.3 times (0.3%) of the main peak area of the control solution, and the peak area of other individual impurities shall not be greater than 0.2 times (0.2%) of the main peak area of the control solution, the sum of each impurity peak area shall not be greater than 0.6 times (0.6%) of the main peak area of the control solution. The peaks in the chromatogram of the test solution which were 0.05 times smaller than the main peak area of the control solution were ignored.
residual solvent
take the right amount of this product, precision weighing, and dimethyl sulfoxide dissolved and quantitatively diluted to make about 0 in each lml. lg of the solution, as the test solution; Another ethanol, dichloromethane, toluene and N, N-dimethylformamide each appropriate amount, precision weighing, A solution containing ethanol 0.5mg, dichloromethane 60UG, toluene 89ug and N, N-dimethylformamide 88ug per 1 ml was prepared by quantitative dilution with dimethyl sulfoxide as a control solution. 5ml of each of the test solution and the reference solution were accurately measured, placed in the headspace bottle, and sealed. Determined according to the residual solvent assay (General 0861 second method). With 6% cyanopropylphenyl-94% dimethylpolysiloxane (or polar similar) as stationary liquid; The initial temperature is 60°C, maintained for 8 minutes, and the temperature is raised to 150°C at a rate of 35°C per minute, maintained for 10 minutes; the inlet temperature was 200°C; The detector temperature was 250°C; The headspace bottle equilibrium temperature was 95°C and the equilibrium time was 60 minutes. Take the reference solution into the headspace, and the separation degree between the peaks of each component shall meet the requirements. The test solution and the reference solution were sampled by Headspace injection, and the chromatograms were recorded. According to the external standard method, the residual amount of ethanol, dichloromethane, toluene and N ,N-dimethylformamide shall be calculated by peak area.
loss on drying
take this product l.O g, dry to constant weight at 105°C, lose no more than 0.5% weight (General rule 0831).
ignition residue
take l.Og of this product and check it according to law (General rule 0841). The residue left shall not exceed 0.1%.
Heavy metals
The residue left under the item of taking the ignition residue shall not contain more than 10 parts per million of heavy metal when examined by law (General Principles 0821, Law II).
145040-37-5 - Content determination
Authoritative Data Verified Data
take this product about 0.45g, precision weighing, add glacial acetic acid-acetic anhydride (1:l)40ml, shake to dissolve, according to the potential titration method (General 0701), with perchloric acid titration solution (0.1 mol/L) titration, and the results of the titration were corrected with a blank test. Each 1 ml of perchloric acid titration solution (0.1 mol/L) corresponds to 61.07mg of C33H34N6O6.
145040-37-5 - Category
Authoritative Data Verified Data
anti-hypertension drugs.
145040-37-5 - Storage
Authoritative Data Verified Data
sealed storage.
Supplier List
CAS: 145040-37-5
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 145040-37-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 145040-37-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 145040-37-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 145040-37-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 145040-37-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 145040-37-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 145040-37-5
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 145040-37-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 145040-37-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 145040-37-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 145040-37-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 145040-37-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 145040-37-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025






