135139-00-3
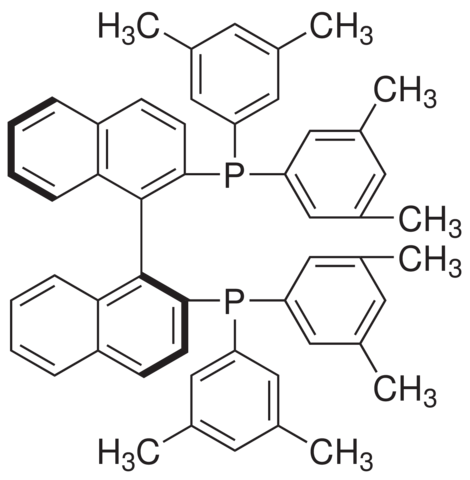
RAC-3,5-Xylyl-Binap
CAS: 135139-00-3;145416-77-9
Molecular Formula: C52H48P2
135139-00-3 - Names and Identifiers
| Name | RAC-3,5-Xylyl-Binap |
| Synonyms | Xylbinap (S)-DM-BINAP (S)-xyl-Binap (S)-xylyl-Binap (S)-3,5-Xyl-BINAP (RAC-Xylyl-Binap) (S)-3,5-Xylyl-Binap (R)-3,5-Xylyl-Binap RAC-3,5-Xylyl-Binap 2,2'-BIS[DI(3,5-XYLYL)PHOSPHINO]-1,1'-BINAPHTHYL Racemic-2,2'-Bis[Di(3,5-Xylyl)phosphino]-1,1'-binaphthyl (S)-(-)-2,2'-Bis[di(3,5-xylyl)phosphino]-1,1'-binaphthyl 2,2'-BIS[BIS(3,5-DIMETHYLPHENYL)PHOSPHINO]-1,1'-BINAPHTHYL RAC-2,2'-Bis(di(3,5-dimethylphenyl)phosphino)-1,1'-binaphthyl 1,1'-Binaphthalene-2,2'-diylbis[bis(3,5-dimethylphenyl)phosphine] 1,1'-binaphthalene-2,2'-diylbis[bis(3,5-dimethylphenyl)phosphane] (S)-(-)-2,2'-Bis[bis(3,5-dimethylphenyl)phosphino]-1,1'-binaphthyl phosphine, 1,1'-[1,1'-binaphthalene]-2,2'-diylbis[1,1-bis(3,5-dimethylphenyl)- (S)-(-)-2,2'-Bis(di-(3,5-dimethylphenyl)phosphino)-1,1'-binaphthyl , ((S)-Xylyl 1,1'-[(1S)-[1,1'-Binaphthalene]-2,2'-diyl]bis[1,1-bis(3,5-dimethylphenyl)-phosphine |
| CAS | 135139-00-3 145416-77-9 |
| EINECS | 681-146-8 |
| InChI | InChI=1/C52H48P2/c1-33-21-34(2)26-43(25-33)53(44-27-35(3)22-36(4)28-44)49-19-17-41-13-9-11-15-47(41)51(49)52-48-16-12-10-14-42(48)18-20-50(52)54(45-29-37(5)23-38(6)30-45)46-31-39(7)24-40(8)32-46/h9-32H,1-8H3 |
135139-00-3 - Physico-chemical Properties
| Molecular Formula | C52H48P2 |
| Molar Mass | 734.89 |
| Melting Point | 203-206°C |
| Boling Point | 825.3±65.0 °C(Predicted) |
| Specific Rotation(α) | -172 º (c=1 in chloroform) |
| Water Solubility | Insoluble in water |
| Appearance | White to beige powder or crystal |
| Color | white to pale yellow |
| Storage Condition | Inert atmosphere,Room Temperature |
| MDL | MFCD01630821 |
135139-00-3 - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37 - Wear suitable gloves. |
| TSCA | No |
135139-00-3 - Reference Information
| application | (S)-binaphthalene (3, 5-xylene) phosphine is a chiral organic phosphine compound, and the metal catalytic reaction it participates in has been widely used in the field of asymmetric synthesis. |
| Synthesis method | Using phosphorus tribromide as raw material, it reacts with bis (3,5-dimethylphenyl) phosphine oxide to obtain bis (3, 5-dimethylphenyl) phosphine bromide in 92% yield, and then it is coupled with Ni(dppe)Cl 2 as catalyst and zinc powder as reducing agent, (S)-binaphthalene (3, 5-xylene) phosphine was obtained. |
Last Update:2024-04-09 21:32:03
Supplier List
Multiple SpecificationsSpot supply
Product Name: (S)-(-)-2,2'-Bis(di-3,5-xylylphosphino)-1,1'-binaphthyl Visit Supplier Webpage Request for quotationCAS: 135139-00-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: (S)-(-)-2,2'-Bis[di(3,5-xylyl)phosphino]-1,1'-binaphthyl Request for quotationCAS: 135139-00-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Multiple SpecificationsSpot supply
Product Name: (S)-(-)-XylBINAP Visit Supplier Webpage Request for quotationCAS: 135139-00-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: (S)-(-)-2,2'-Bis(di-3,5-xylylphosphino)-1,1'-binaphthyl Visit Supplier Webpage Request for quotationCAS: 135139-00-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: (S)-(-)-2,2'-Bis[di(3,5-xylyl)phosphino]-1,1'-binaphthyl Request for quotationCAS: 135139-00-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Multiple SpecificationsSpot supply
Product Name: (S)-(-)-XylBINAP Visit Supplier Webpage Request for quotationCAS: 135139-00-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


