13362-28-2
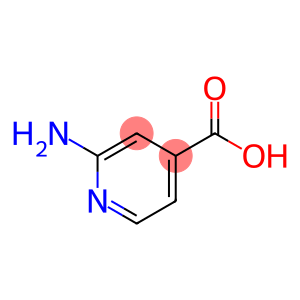
2-Aminoisonicotinic acid
CAS: 13362-28-2
Molecular Formula: C6H6N2O2
13362-28-2 - Names and Identifiers
| Name | 2-Aminoisonicotinic acid |
| Synonyms | CHEMBRDG-BB 4015121 2-AMINOISONICOTINIC ACID 2-Aminoisonicotinic acid 2-AMINOPYRIDINE-4-CARBOXYLATE 2-AMINO-4-PYRIDINECARBOXYLIC ACID 2-Aminopyridine-4-carboxylic acid 2-AMINOPYRIDINE-4-CARBOXYLIC ACID 2-Amino-4-Pyridine Carboxylic Acid 4-Pyridinecarboxylicacid,2-amino-(9CI) 2-AMINO-4-PYRIDINECARBOXYLIC ACID AMIDE |
| CAS | 13362-28-2 |
| EINECS | 681-100-7 |
| InChI | InChI=1/C6H6N2O2/c7-5-3-4(6(9)10)1-2-8-5/h1-3H,(H2,7,8)(H,9,10) |
| InChIKey | QMKZZQPPJRWDED-UHFFFAOYSA-N |
13362-28-2 - Physico-chemical Properties
| Molecular Formula | C6H6N2O2 |
| Molar Mass | 138.12 |
| Density | 1.417±0.06 g/cm3(Predicted) |
| Melting Point | 300-302°C |
| Boling Point | 498.5±30.0 °C(Predicted) |
| Flash Point | 255.3°C |
| Vapor Presure | 9.33E-11mmHg at 25°C |
| Appearance | Solid |
| Color | Off-white to brown |
| BRN | 3156 |
| pKa | 2.11±0.10(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Refractive Index | 1.649 |
| MDL | MFCD00233714 |
13362-28-2 - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R23/24/25 - Toxic by inhalation, in contact with skin and if swallowed. R22 - Harmful if swallowed |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| WGK Germany | 3 |
| HS Code | 29339900 |
| Hazard Note | Irritant/Keep Cold |
| Hazard Class | IRRITANT |
13362-28-2 - Reference Information
| introduction | 2-amino isonicotinic acid is a common organic synthesis intermediate, and its appearance is light yellow crystal at normal temperature and pressure. The molecular structure of 2-aminoisonicotinic acid contains both basic amino groups and acidic carboxyl groups; therefore, in its preparation, separation and preparation process must pay attention to adjust the pH of the reaction system, which is generally adjusted The conditions for neutral partial acid. |
| Use | 2-Amino isonicotinic acid is a common organic synthesis intermediate, and its main use is to use it as a molecular skeleton for molecular structure modification and synthesis of pyridine-containing drugs. In addition, because the compound contains both amino groups and carboxyl groups, the molecule also has a good application in supramolecular chemistry and the construction of metal organic frameworks. The carboxyl group on the pyridine ring can be converted into ester group under general acidic esterification conditions, or converted into amide by dichlorosulfoxide plus amine. |
| Synthesis method | Starting from 2-chloroisonicotinic acid through the nucleophilic substitution reaction of ammonia water is a more common synthesis method. However, the main disadvantage of this route is that it requires a high reaction temperature and has stricter requirements on the reaction equipment. At the same time, after the reaction, attention must be paid to adjusting the pH of the reaction system, which is generally adjusted to neutral partial acid. |
| environmental hazards | 2-amino isonicotinic acid, as an organic compound containing halogen-containing heteroaromatic ring, is more harmful to the water environment, and undiluted or large amounts of products cannot be allowed to contact groundwater, waterways or sewage systems. |
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: 2-Aminoisonicotinic acid Visit Supplier Webpage Request for quotationCAS: 13362-28-2
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2-Aminopyridine-4-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 13362-28-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Aminoisonicotinic acid Request for quotation
CAS: 13362-28-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13362-28-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple Specifications
Product Name: 2-Aminoisonicotinic acid Visit Supplier Webpage Request for quotationCAS: 13362-28-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Aminoisonicotinic acid Visit Supplier Webpage Request for quotationCAS: 13362-28-2
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2-Aminopyridine-4-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 13362-28-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Aminoisonicotinic acid Request for quotation
CAS: 13362-28-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13362-28-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple Specifications
Product Name: 2-Aminoisonicotinic acid Visit Supplier Webpage Request for quotationCAS: 13362-28-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



