13360-61-7
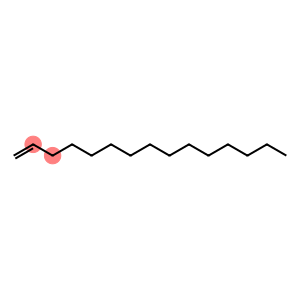
pentadec-1-ene
CAS: 13360-61-7
Molecular Formula: C15H30
13360-61-7 - Names and Identifiers
| Name | pentadec-1-ene |
| Synonyms | Pentadecene 1-PENTADECENE pentadec-1-ene Pentadec-1-ene Pentadecene,1- |
| CAS | 13360-61-7 |
| EINECS | 236-414-5 |
| InChI | InChI=1/C15H30/c1-3-5-7-9-11-13-15-14-12-10-8-6-4-2/h3H,1,4-15H2,2H3 |
13360-61-7 - Physico-chemical Properties
| Molecular Formula | C15H30 |
| Molar Mass | 210.4 |
| Density | 0.775g/mLat 25°C(lit.) |
| Melting Point | −4°C(lit.) |
| Boling Point | 268-269°C(lit.) |
| Flash Point | >230°F |
| Water Solubility | Not miscible or difficult to mix in water. |
| Vapor Presure | 0.0125mmHg at 25°C |
| Appearance | clear liquid |
| Specific Gravity | 0.775 |
| Color | Colorless |
| BRN | 1751011 |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | n20/D 1.439(lit.) |
| MDL | MFCD00008987 |
13360-61-7 - Risk and Safety
| Safety Description | S23 - Do not breathe vapour. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| TSCA | Yes |
13360-61-7 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| application | pentadecene is an olefin. Olefins are a very important type of synthetic intermediates in organic chemistry. Various other groups can be easily introduced through the reaction of olefins, such as the preparation of halogenates through addition reactions, and the preparation of cyclic compounds through D-A reactions; on the other hand, olefins can also undergo various coupling reactions through Heck reaction, Suzuki reaction, etc. It is widely used in the field of synthesis. |
| preparation | adding photocatalyst palladium chloride (2mol%,0.7mg), 4,5-diphenylphosphine -9, 9-dimethyloxanthracene (L1)(3mol%) to 10mL of Schlenk reaction tube (F891410 reaction tube, capacity 10mL, grinding port 14/20), 3.5mg), 2-(dicyclohexylphosphinyl) biphenyl (L2)(4mol%,2.8mg) and active aliphatic NHPI ester (0.2mmol,80.2mg). The air in the tube was completely replaced three times with argon, and then 2,4, 6-trimethylpyridine (0.2mmol,24.2mg) was added in argon atmosphere, and N,N-dimethylacetamide (DMA) was 2mL. The reaction system was continuously stirred for 15 hours at room temperature under 36W blue LED lamp irradiation (IKA magnetic stirrer, RCT basic type, stirring speed 500 r/min minutes). After the reaction is completed, the reaction is quenched with H2O, and the reaction solution is extracted with ethyl acetate (3*10mL), and the combined organic phase is concentrated by rotary evaporation (BUCHI rotary evaporator R-3). The concentrated residue was separated by chromatography on a chromatographic column (C383040C storage ball chromatography column with sand plate, 35/20, effective length: 500mm) to obtain the product. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 1-pentadecene Visit Supplier Webpage Request for quotationCAS: 13360-61-7
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 1-Pentadecene Visit Supplier Webpage Request for quotationCAS: 13360-61-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-PENTADECENE Request for quotation
CAS: 13360-61-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13360-61-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 1-Pentadecene Visit Supplier Webpage Request for quotation
CAS: 13360-61-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 13360-61-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1-pentadecene Visit Supplier Webpage Request for quotationCAS: 13360-61-7
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 1-Pentadecene Visit Supplier Webpage Request for quotationCAS: 13360-61-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-PENTADECENE Request for quotation
CAS: 13360-61-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13360-61-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 1-Pentadecene Visit Supplier Webpage Request for quotation
CAS: 13360-61-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 13360-61-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



