124937-51-5
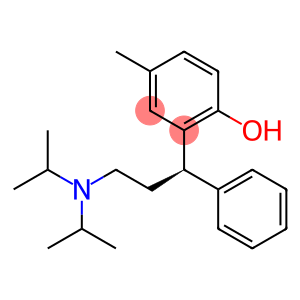
Tolterodine
CAS: 124937-51-5
Molecular Formula: C22H31NO
124937-51-5 - Names and Identifiers
| Name | Tolterodine |
| Synonyms | DETROL Kabi 2234 PNU 200583 DETRUSITOL Tolterodine 124937-51-5 (R)-Tolterodine R-(+)-Tolterodine 2-[3-(dipropan-2-ylamino)-1-phenylpropyl]-4-methylphenol 2-{3-[bis(1-methylethyl)amino]-1-phenylpropyl}-4-methylphenol 2-[(1S)-3-(dipropan-2-ylamino)-1-phenylpropyl]-4-methylphenol phenol, 2-[3-[bis(1-methylethyl)amino]-1-phenylpropyl]-4-methyl- 2-[(1R)-3-[Bis(1-methylethyl)amino]-1-phenylpropyl]-4-methyl-phenol 2-[3-(diisopropylamino)-1-phenyl-propyl]-4-methyl-phenol hydrochloride (R)-(+)-N,N-Diisopropyl-3-(2-hydroxy-5-methylphenyl)-3-phenylpropylamine |
| CAS | 124937-51-5 |
| InChI | InChI=1/C22H31NO.ClH/c1-16(2)23(17(3)4)14-13-20(19-9-7-6-8-10-19)21-15-18(5)11-12-22(21)24;/h6-12,15-17,20,24H,13-14H2,1-5H3;1H |
124937-51-5 - Physico-chemical Properties
| Molecular Formula | C22H31NO |
| Molar Mass | 325.49 |
| Density | 1.003±0.06 g/cm3(Predicted) |
| Boling Point | 442.2±45.0 °C(Predicted) |
| Specific Rotation(α) | D25 +72° (c = 1.0 in CH2Cl2) |
| Flash Point | 237.5°C |
| Solubility | DMSO: ≥20mg/mL |
| Vapor Presure | 2.02E-09mmHg at 25°C |
| Appearance | powder |
| Color | white to off-white |
| pKa | pKa 9.8 (Uncertain) |
| Storage Condition | -20°C Freezer |
124937-51-5 - Risk and Safety
| Risk Codes | R63 - Possible risk of harm to the unborn child R51/53 - Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S57 - Use appropriate container to avoid environmental contamination. |
| WGK Germany | 3 |
124937-51-5 - Use
Open Data Verified Data
developed by Pharmacia & Upiohn, Inc., USA, which was first launched in Sweden in February 1998 and in the United Kingdom and the United States in the same year. It was launched in France in April 1999. Muscarinic receptor antagonists. For the treatment of bladder stimulation caused by urinary incontinence, urinary frequency and urgency and other symptoms.
Last Update:2024-01-02 23:10:35
124937-51-5 - Safety
Open Data Verified Data
male mice were injected intravenously with LDso: 10~20mg/kg.
Last Update:2022-01-01 09:24:25
124937-51-5 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 3.072 ml | 15.362 ml | 30.723 ml |
| 5 mM | 0.614 ml | 3.072 ml | 6.145 ml |
| 10 mM | 0.307 ml | 1.536 ml | 3.072 ml |
| 5 mM | 0.061 ml | 0.307 ml | 0.614 ml |
Last Update:2024-01-02 23:10:35
124937-51-5 - Reference Information
| Biological activity | Tolterodine(PNU-200583) is a muscarinic receptor (mAChR) antagonist. |
| Use | Tolterodine belongs to a class of drugs called antimuscarinic, used to treat overactive bladder (the bladder muscles contract uncontrollably and cause frequent urination, Urgent need to urinate and unable to control urination), it works by relaxing the bladder muscles to prevent bladder contraction. |
| production method | trans-internal cinnamic acid (100g,675 mmo1) is added to the reaction bottle, p-cresol (76.6g,708 mmo1) preheated to 60 ℃ is added, and then concentrated sulfuric acid (13.0ml,243 mmo1) is added. After addition, immediately heat to 122.5 ℃, then at 120-125 ℃; Stir until the reaction is complete, about 6 hours. Cool to 100 ℃ and pour into the preheated separatory funnel. The lower acid layer was separated and 280ml of toluene, 50ml of water and 10ml of 47% potassium carbonate solution were added. If necessary, 47% potassium carbonate can be used to adjust the Ph value of the water layer to 5~8. The organic layer is separated, washed with 50ml of water, and concentrated to about 150ml under reduced pressure. Add 350ml isopropanol and concentrate until 350ml remains. Add 150ml isopropanol and concentrate until 350ml remains. Add another 150ml of isopropanol and concentrate until 350ml remains. Cool to 30-40°C under rapid stirring to form crystals, and continue stirring. Then cool to 0~5 ℃ and keep for 1h. Filter to collect crystals, and wash in batches with 200ml isopropanol at 0~5 ℃. If the final lotion still has color, continue washing until the lotion is colorless. Dry at 60 ℃ and under reduced pressure to obtain 3, 4-dihydro-6-methyl-4-phenyl-2H-benzopyran-2-one with a melting point of 83~85 ℃. 3, 4-dihydro-6-methyl-4-phenyl-2H-benzopyran-2-one (100.0g,420.2 mmo1) was added to 500ml of toluene, cooled to -21 ℃ and under nitrogen protection, slowly added to toluene solution of diisobutylaluminum hydrogen (DIBAL,1.5mol/L,290ml,435 mmo1) in 2h, and maintain the reaction temperature at -20 ~-25 ℃. When the reaction is complete, 45ml of ethyl acetate is added at -20-25°C. Then 500ml of 23% citric acid was added, and the solution was stirred at 45-50 ℃ for lh (or overnight at 20-25 ℃). The organic layer is separated, washed with 2 × 300ml of water, and concentrated to 250ml under reduced pressure. Add 500ml of methanol, concentrate to 250ml, repeat the operation. Then concentrate to oil, let it stand and crystallize to obtain 3, 4-dihydro-6-methyl-4-phenyl-2H-benzopyran-2-ol, which is directly used in the next reaction. 100g of 3,4-dihydro-6-methyl-4-phenyl-2H-benzopyran-2-ol was slowly added with 22g of 5% palladium-carbon (1.5 mmo1) under the protection of nitrogen in a solution of 500ml of methanol. Diisopropylamine (147.0ml,1.05 mo1) was added and hydrogenated to complete at 0.32~0.35MPa and 48 ℃. Usually the reaction is about 10h, or it can be reacted overnight. Cool, remove the reactor, and then clean the reactor with 150ml of methanol. The cleaning solution and the reaction solution are combined, filtered, the filtrate is concentrated, and then ethyl acetate is added to 700ml, and heated to 55 ℃. In 15min, 52.5ml concentrated hydrochloric acid was added. Slowly cool to -15-20°C and keep it for 1h. Filtration to collect solids and vacuum drying overnight to obtain tolteridine hydrochloride with a melting point of 199~201 ℃. |
Last Update:2024-04-09 02:00:06
Supplier List
Spot supply
Product Name: Tolterodine Visit Supplier Webpage Request for quotationCAS: 124937-51-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Tolterodine Visit Supplier Webpage Request for quotationCAS: 124937-51-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Tolterodine Request for quotation
CAS: 124937-51-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 124937-51-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Tolterodine Visit Supplier Webpage Request for quotation
CAS: 124937-51-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 124937-51-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Tolterodine Visit Supplier Webpage Request for quotationCAS: 124937-51-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Tolterodine Visit Supplier Webpage Request for quotationCAS: 124937-51-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Tolterodine Request for quotation
CAS: 124937-51-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 124937-51-5
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Tolterodine Visit Supplier Webpage Request for quotation
CAS: 124937-51-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 124937-51-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



