119006-77-8
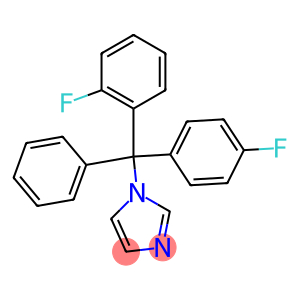
Flutrimazole
CAS: 119006-77-8
Molecular Formula: C22H16F2N2
119006-77-8 - Names and Identifiers
| Name | Flutrimazole |
| Synonyms | Micetal flutrimazol Flutrimazole FLUTRIMAZOLC FLUTRIMAZOLE 1-[o-Fluoro-a-(p-fluorophenyl)-a-phenylbenzyl]imidazole 1-[(2-Fluorophenyl)(4-fluorophenyl)(phenyl)methyl]imidazole 1-((2-fluorophenyl)(4-fluorophenyl)phenylmethyl)-1h-imidazol 1-[(2-Fluorophenyl)(4-fluorophenyl)phenylmethyl]-1H-imidazole 1-[(2-Fluorophenyl)-(4-fluorophenyl)phenylmethyl]-1H-imidazole 1-[(2-fluorophenyl)-(4-fluorophenyl)phenylmethyl]-1h-imidazole 1-(o-fluoro-alpha-(p-fluorophenyl)-alpha-phenylbenzyl)imidazole |
| CAS | 119006-77-8 |
| InChI | InChI=1/C22H16F2N2/c23-19-12-10-18(11-13-19)22(26-15-14-25-16-26,17-6-2-1-3-7-17)20-8-4-5-9-21(20)24/h1-16H |
119006-77-8 - Physico-chemical Properties
| Molecular Formula | C22H16F2N2 |
| Molar Mass | 346.37 |
| Density | 1.15±0.1 g/cm3(Predicted) |
| Melting Point | 161-166℃ |
| Boling Point | 457.1±40.0 °C(Predicted) |
| Flash Point | 230.3°C |
| Solubility | Practically insoluble in water, freely soluble in tetrahydrofuran, soluble in methanol. |
| Vapor Presure | 4.16E-08mmHg at 25°C |
| Appearance | neat |
| Color | White to Pale Yellow |
| pKa | 6.03±0.34(Predicted) |
| Storage Condition | -20°C Freezer |
| Refractive Index | 1.59 |
| Physical and Chemical Properties | Off-white powder to white crystalline powder |
119006-77-8 - Risk and Safety
| Toxicity | LD50 in male, female mice, male, female rats (mg/kg): >1000, >1000, 808, 1214 orally; >2000, >2000, 1079, 1446 i.p. (Vericat) |
119006-77-8 - Reference Information
| imidazole antifungal | flutramazole is a new type of topical imidazole antifungal drug with broad-spectrum antibacterial activity. it was developed by Spain's Urich pharmaceutical company. it was first listed in Spain in 1995 and then listed in Chile, Peru, Argentina, south Korea, Mexico, Greece, Italy and other countries, formulations used in the market include creams, gels and solutions. It is suitable for the topical treatment of epidermal mold infections caused by yellow ringworm, microsporum canis and epidermophyton flocculus, such as tinea manus, tinea pedis, tinea pedis, tinea corporis, tinea versicolor, tinea capitis, onychomycosis, facial tinea, tinea cruris and vulvitis. It is also suitable for Candida epidermis infection caused by Candida yeast and pityriasis versicolor. The mechanism of action is to inhibit the biosynthesis of ergosterol on the fungal cell membrane by inhibiting the activity of fungal cytochrome P-450, damage the fungal cell membrane and change its permeability, thereby inhibiting the growth of fungi. Its IC50 value is 0.071 μmol/L. It has a strong inhibitory effect on dermatophytes, filamentous fungi and yeasts that cause saprophytic and pathogenic diseases in animals and humans. The minimum inhibitory concentration of most test strains is 0.025-5.0 μg/ml. Its curative effect is definite and its side effect is small. Flutrimazole (MIC = 0.15-0.6 mg/ml) has the same inhibitory effect on fungi as clotrimazole (MIC = 0.3-2.5 mg/ml), better than ketoconazole, and significantly higher than bifonazole (the difference in minimum inhibitory concentration is greater than 1 order of magnitude). It has certain local anti-inflammatory activity, good tolerance, rare local irritation and allergy, and is a safe and effective drug. |
| use | flutramazole is a new type of topical imidazole antifungal drug with broad-spectrum antibacterial activity and was developed by Spanish Urich pharmaceutical company. |
| usage and dosage | once a day for adults and children over 10 years old. Apply an appropriate amount of ointment to the damaged part and the surrounding skin, and gently rub it to facilitate drug penetration. Use a small amount of ointment to avoid softening of the skin in the damaged area with wrinkles. The course of treatment should be determined according to the degree of damage or the type of microorganisms infected and the site of infection. Generally speaking, in the first few days of use, the skin symptoms can be significantly improved. The recommended course of treatment to reduce recurrence is: tinea pedis and interphalangeal mycotis, 4 weeks; tinea corporis, 2 to 3 weeks; pityriasis versicolor, 1 to 2 weeks; Candida epidermis infection, 2 to 4 weeks. If the clinical effect is not obvious after 4 weeks of treatment, it needs to be rediagnosed. It is recommended that patients pay attention to daily hygiene to control the possible source of infection and recurrence. |
| synthesis method | o-fluorobenzoyl chloride reacts with benzoate-gram, grigni reaction with p-bromofluorobenzene, methanesulfonyl chloride sulfonylation, and finally substitution reaction with imidazole to prepare the antifungal drug flutramazole with a total yield of 59%. |
| toxicity | mice oral LD50>1000mg/Kg, intraperitoneal injection LD50>2000mg/Kg. The LD50 of male rats was 808mg/Kg and 1079mg/Kg by oral and intraperitoneal injection, respectively. The LD50 of female rats was 1214mg/Kg and 1446mg/Kg by oral and intraperitoneal injection, respectively. The results of long-term toxicity test showed that there was no significant difference between flutrimazole and placebo, and there was no carcinogenic, teratogenic and mutagenic activity. |
| intellectual property and patent | the original compound patent of flutramazole is 1990. the European patent number is EP352352, and the applicant is Urich pharmaceutical company of Spain. However, there is no patent in China, so there is no intellectual property problem in the development of flutrimazole and flutrimazole cream in China. |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 1-[(2-Fluorophenyl)(4-fluorophenyl)(phenyl)methyl]imidazole Visit Supplier Webpage Request for quotationCAS: 119006-77-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Flutrimazole Request for quotation
CAS: 119006-77-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 119006-77-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple Specifications
Product Name: Flutrimazole Visit Supplier Webpage Request for quotationCAS: 119006-77-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1-[(2-Fluorophenyl)(4-fluorophenyl)(phenyl)methyl]imidazole Visit Supplier Webpage Request for quotationCAS: 119006-77-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Flutrimazole Request for quotation
CAS: 119006-77-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 119006-77-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple Specifications
Product Name: Flutrimazole Visit Supplier Webpage Request for quotationCAS: 119006-77-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


