116855-08-4
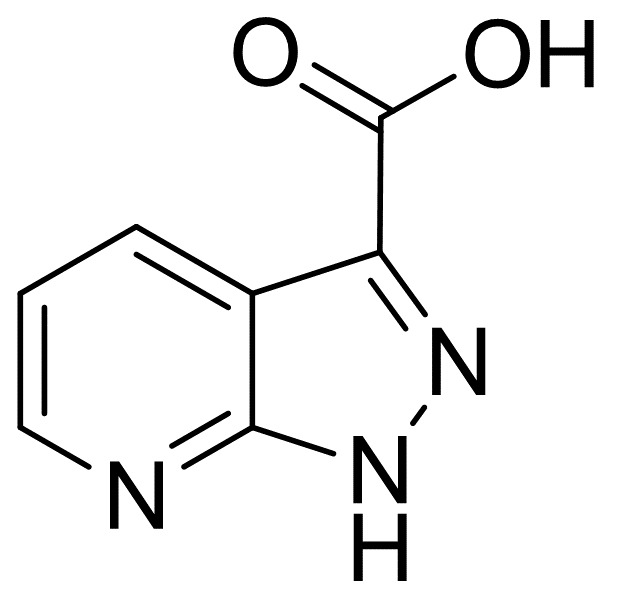
1H-Pyrazolo[3,4-b]pyridine-3-carboxylicacid
CAS: 116855-08-4
Molecular Formula: C7H5N3O2
116855-08-4 - Names and Identifiers
| Name | 1H-Pyrazolo[3,4-b]pyridine-3-carboxylicacid |
| Synonyms | 1H-Pyrazolo[3,4-b]pyridin-3-carboxylic acid 1H-Pyrazolo[3,4-b]pyridine-3-carboxylicacid 1H-pyrazolo[3,4-b]pyridine-3-carboxylic acid 1H-Pyrazolo[3,4-b]pyridine-3-carboxylic acid 2h-pyrazolo[3,4-b]pyridine-3-carboxylic Acid 1H-Pyrazolo[3,4-b]pyridine-3-carboxylicacid(9CI) |
| CAS | 116855-08-4 |
| InChI | InChI=1/C7H5N3O2/c11-7(12)5-4-2-1-3-8-6(4)10-9-5/h1-3H,(H,11,12)(H,8,9,10) |
116855-08-4 - Physico-chemical Properties
| Molecular Formula | C7H5N3O2 |
| Molar Mass | 163.13 |
| Density | 1.68 |
| Melting Point | 265-270℃ (DEC.) |
| Boling Point | 391.1±25.0 °C(Predicted) |
| Flash Point | 241.107°C |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | Solid |
| pKa | 8.83±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.763 |
| MDL | MFCD11040725 |
116855-08-4 - Risk and Safety
| Risk Codes | 20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S24/25 - Avoid contact with skin and eyes. S36/37 - Wear suitable protective clothing and gloves. |
| HS Code | 29339900 |
116855-08-4 - Reference Information
| Use | 1H-pyrazolyl [3,4-B] pyridine-3-carboxylic acid is a carboxylic acid derivative, it can be used as an intermediate in the synthesis of medicine and in the experimental research of medicine. |
| preparation | at room temperature, A mass of 2-chloro-3-acetylpyridine was added to the condenser tube and an electrically stirred three-necked flask, and hydrazine hydrate was added as a solvent. The reaction was stirred in organic solvent for 1 hour, heated to reflux overnight, and the reaction progress was detected by thin layer chromatography. When the reaction was finished, the temperature was stopped, and the system was cooled to room temperature, then purified water and dichloromethane were added, the reaction solution was extracted, and the organic phase was collected after standing and stratification, and the aqueous phase was washed many times. After the organic phase was combined, anhydrous sodium sulfate was added to dry, and the organic phase was concentrated and purified by silica gel column chromatography, the compound 1H-pyrazolyl [3,4-B] pyridine-3-carboxylic acid was obtained. |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 1H-Pyrazolo[3,4-b]pyridine-3-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 116855-08-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1H-Pyrazolo[3,4-b]pyridine-3-carboxylicacid(9CI) Request for quotation
CAS: 116855-08-4
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 116855-08-4
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 1H-Pyrazolo[3,4-b]pyridine-3-carboxylic acid Visit Supplier Webpage Request for quotation
CAS: 116855-08-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 116855-08-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1H-Pyrazolo[3,4-b]pyridine-3-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 116855-08-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1H-Pyrazolo[3,4-b]pyridine-3-carboxylicacid(9CI) Request for quotation
CAS: 116855-08-4
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 116855-08-4
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 1H-Pyrazolo[3,4-b]pyridine-3-carboxylic acid Visit Supplier Webpage Request for quotation
CAS: 116855-08-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 116855-08-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


