10294-38-9
Barium chlorate
CAS: 10294-38-9
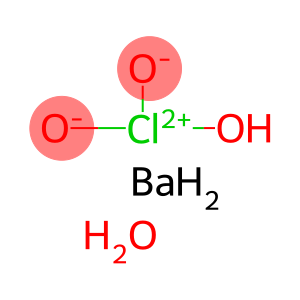
Molecular Formula: BaClH5O4
10294-38-9 - Names and Identifiers
| Name | Barium chlorate |
| Synonyms | Barium chlorate BariumchlorateH2O Barium dichlorate BARIUM CHLORATE 1-HYDRATE CHLORANILIC ACID, BARIUM SALT BARIUM CHLORANILATE TRIHYDRATE BARIUM CHLOROANILATE TRIHYDRATE CHLORANILIC ACID BARIUM SALT TRIHYDRATE BARIUMCHLORATEMONOHYDRATE,CRYSTAL,REAGENT 2,5-DICHLORO-3,6-DIHYDROXY-1,4-BENZOQUINONE BARIUM SALT TRIHYDRATE |
| CAS | 10294-38-9 |
| EINECS | 680-417-8 |
| InChI | InChI=1/Ba.2ClHO3.H2O/c;2*2-1(3)4;/h;2*(H,2,3,4);1H2/q+2;;;/p-2 |
10294-38-9 - Physico-chemical Properties
| Molecular Formula | BaClH5O4 |
| Molar Mass | 241.81 |
| Density | 3.18g/mLat 25°C(lit.) |
| Melting Point | 414°C(lit.) |
| Water Solubility | g/100g H2O: 20.3 (0°C), 26.9 (10°C), 33.9 (20°C), 41.6 (30°C), 49.7 (40°C), 66.7 (60°C), 84.8 (80°C), 105 (100°C); soluble HCl [LAN05] [MER06] |
| Appearance | white monoclinic crystals |
| Exposure Limit | ACGIH: TWA 0.5 mg/m3NIOSH: IDLH 50 mg/m3; TWA 0.5 mg/m3 |
| Use | Used as an analytical reagent and also used in the pharmaceutical industry |
10294-38-9 - Risk and Safety
| Risk Codes | R20/22 - Harmful by inhalation and if swallowed. R51/53 - Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R9 - Explosive when mixed with combustible material |
| Safety Description | S28 - After contact with skin, wash immediately with plenty of soap-suds. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S27 - Take off immediately all contaminated clothing. S13 - Keep away from food, drink and animal foodstuffs. |
| UN IDs | UN 1564 6.1/PG 3 |
| WGK Germany | 1 |
| TSCA | Yes |
| Hazard Class | 5.1 |
| Packing Group | II |
10294-38-9 - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Introduction | barium chlorate monohydrate is an inorganic compound with white crystals. It can be used as an analytical reagent and also in the pharmaceutical industry. |
| physical properties | barium chlorate monohydrate colorless monoclinic system, losing crystal water at 120 degrees. Oxygen was released at 250 degrees Celsius. Soluble in water, solubility (anhydrous salt meter) 20 degrees 25 grams, 100 degrees 52. Insoluble in ethanol or propanol. When mixed with a combustible material, the explosion occurs on heating or impact. |
| preparation | barium chlorate monohydrate is prepared by electrolytic oxidation of barium chloride. The method and principle are similar to that of preparation of sodium chlorate by electrolysis. |
| Application | barium chlorate monohydrate is used for making explosives, matches and pyrotechnic products, and is also used as a mordant. |
| Use | used as an analytical reagent, and also used in the pharmaceutical industry |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Barium chlorate monohydrate 98% Visit Supplier Webpage Request for quotationCAS: 10294-38-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Barium chlorate monohydrate 98% Visit Supplier Webpage Request for quotationCAS: 10294-38-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
View History
