100-88-9
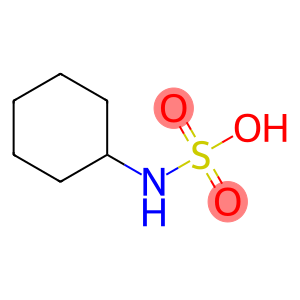
N-cyclohexylsulfamic acid
CAS: 100-88-9
Molecular Formula: C6H13NO3S
100-88-9 - Names and Identifiers
100-88-9 - Physico-chemical Properties
| Molecular Formula | C6H13NO3S |
| Molar Mass | 179.24 |
| Density | 1.149 (estimate) |
| Melting Point | ~180°C (dec.) |
| Boling Point | 332.48℃[at 101 325 Pa] |
| Water Solubility | 133g/L |
| Solubility | dioxane: soluble1g/10 mL, clear, colorless (hot) |
| Vapor Presure | 0Pa at 25℃ |
| Appearance | White crystal |
| Color | White |
| Merck | 14,2703 |
| BRN | 2208885 |
| pKa | pKa 2.28(H2O t=RT c=0.056) (Uncertain) |
| Storage Condition | Refrigerator |
| Refractive Index | 1.5500 (estimate) |
| MDL | MFCD00065234 |
| Physical and Chemical Properties | Colorless crystal or white crystalline powder with sweet and sour taste. |
100-88-9 - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 2 |
| RTECS | GV6950000 |
| HS Code | 2929909000 |
100-88-9 - Reference Information
| LogP | -1.61 at 25℃ |
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| identification test | solubility (OT-42) soluble in water and ethanol. Precipitation test to prepare 2% sample solution, acidify with hydrochloric acid, add lml of barium chloride test solution (TS-37), and filter if turbidity or precipitation occurs. Add 10% sodium nitrite solution lml to the clarified solution. There should be white precipitation. |
| content analysis | accurately weigh the sample about 350mg and move it into a 250ml flask. Add 50ml of water to dissolve it, add phenolphthalein test solution (TS-167), and titrate with 0.1mol/L sodium hydroxide solution. 0.1mol/L sodium hydroxide solution per ml is equivalent to 17.82mg of C6H13NO3S. |
| toxicity | ADI has not yet been specified (FAO/WHO,2001). |
| use | non-nutritious sweetener. |
| Production method | It is formed by sulfonating cyclohexylamine with chlorosulfonic acid or sulfamate. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 19:55:45
Supplier List
Spot supply
Product Name: N-Cyclohexylsulfamic acid Visit Supplier Webpage Request for quotationCAS: 100-88-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Cyclamic acid Request for quotation
CAS: 100-88-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 100-88-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: N-?Cyclohexylsulfamic Acid (Cyclamic Acid) Visit Supplier Webpage Request for quotationCAS: 100-88-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: N-Cyclohexylsulfamic acid Visit Supplier Webpage Request for quotationCAS: 100-88-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Cyclamic acid Request for quotation
CAS: 100-88-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 100-88-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: N-?Cyclohexylsulfamic Acid (Cyclamic Acid) Visit Supplier Webpage Request for quotationCAS: 100-88-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


