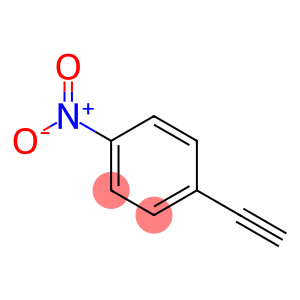
1-ethynyl-4-nitrobenzene
1-ethynyl-4-nitrobenzene
CAS: 937-31-5
Molecular Formula: C8H5NO2
1-ethynyl-4-nitrobenzene - Names and Identifiers
| Name | 1-ethynyl-4-nitrobenzene |
| Synonyms | NSC 71089 (4-Nitrophenyl)ethyne p-Ethynylnitrobenzene 4-Ethynylnitrobenzene 4-Nitroethynylbenzene 4-Nitrophenylacetylene (p-Nitrophenyl)acetylene 1-ethynyl-4-nitrobenzene 4-Ethynyl-1-nitrobenzene 1-Nitro-4-ethynylbenzene |
| CAS | 937-31-5 |
| InChI | InChI=1/C8H5NO2/c1-2-7-3-5-8(6-4-7)9(10)11/h1,3-6H |
1-ethynyl-4-nitrobenzene - Physico-chemical Properties
| Molecular Formula | C8H5NO2 |
| Molar Mass | 147.13 |
| Density | 1.22±0.1 g/cm3(Predicted) |
| Melting Point | 148-150°C(lit.) |
| Boling Point | 246.6±23.0 °C(Predicted) |
| Flash Point | 112.5°C |
| Solubility | Ethanol, Ether |
| Vapor Presure | 0.0423mmHg at 25°C |
| Appearance | Solid |
| Color | Light Yellow |
| Maximum wavelength(λmax) | ['286nm(CH2Cl2)(lit.)'] |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.579 |
| MDL | MFCD00024794 |
1-ethynyl-4-nitrobenzene - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| HS Code | 29049090 |
1-ethynyl-4-nitrobenzene - Reference Information
| Application | p-nitrophenyl acetylene is an intermediate in organic synthesis and pharmaceutical intermediates, which can be used in laboratory research and development process and chemical and pharmaceutical synthesis process. |
| preparation | step 1 [4-(nitrophenyl) ethynyl] trimethylsilane (S7g) preparation of 1-iodo-4-nitrobenzene (S6)(1.00g,4.02 mmol),Pd(PPh3)2 Cl2(84.3 mg,0.120 mmol),CuI(45.7) (Trimethylsilyl) acetylene (2.24 mg,16.4 mmol)(S5) was added to a solution of Et3N (560 mg,4.06 mmol) in 6.8ml DMF. After stirring at 50 °c for 1 H, the reaction mixture was diluted with AcOEt and H2O. The organic layer was washed three times with water, washed with brine, dried over Na2SO4, filtered, and concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (hexane/AcOEt = 2:1) to give [4-(nitrophenyl) ethynyl]-trimethylsilane S7g. Yellow solid, 730mg,3.33mmol,83%. 1 H NMR(400 MHz,CDCl3) Δ8.17 (d,J = 8.8Hz,2H),7.59(d,J = 8.8Hz,2H),0.28(s,9H). Step 2 4-nitrophenylacetylene (8g) to [4-(nitrophenyl) ethynyl] trimethylsilane S7g(69.9mg,0.318mmol) in THF(0.43mL) at 0 °c TBAF(1.0m in THF, 0.38mL,38 μmol) was added to the solution. After stirring for 15 min, the reaction was quenched with saturated aqueous NaHCO3 and the resulting mixture was extracted three times with Et2O. The combined organic extracts were washed with brine, dried over Na2SO4, filtered, and concentrated under reduced pressure. The residue was purified by silica gel flash column chromatography (hexane/AcOEt = 1:10) and gel permeation chromatography to give 8g of alkyne. White solid, 37.5mg,0.255mmol,80%. |
| Use | 4-nitrophenylacetylene is an organic intermediate, it can be prepared from 1-iodo-4-nitrobenzene in two steps. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 4-NITROPHENYLACETYLENE Request for quotation
CAS: 937-31-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 937-31-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 1-ETHYNYL-4-NITROBENZENE Visit Supplier Webpage Request for quotationCAS: 937-31-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 1-Ethynyl-4-Nitrobenzene Visit Supplier Webpage Request for quotationCAS: 937-31-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 4-NITROPHENYLACETYLENE Request for quotation
CAS: 937-31-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 937-31-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 1-ETHYNYL-4-NITROBENZENE Visit Supplier Webpage Request for quotationCAS: 937-31-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 1-Ethynyl-4-Nitrobenzene Visit Supplier Webpage Request for quotationCAS: 937-31-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


