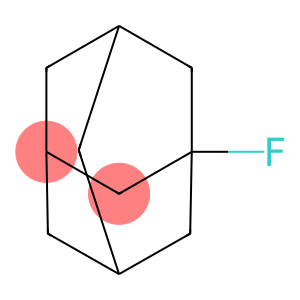
1-Fluoroadamantane
1-Fluoroadamantane
CAS: 768-92-3
Molecular Formula: C10H15F
1-Fluoroadamantane - Names and Identifiers
| Name | 1-Fluoroadamantane |
| Synonyms | 1-FLUOROADAMANTANE 1-Fluoroadamantane 1-ADAMANTYL FLUORIDE 1-fluorotricyclo[3.3.1.1~3,7~]decane Tricyclo[3.3.1.13,7]decane, 1-fluoro- tricyclo[3.3.1.1~3,7~]decane, 1-fluoro- |
| CAS | 768-92-3 |
| InChI | InChI=1/C10H15F/c11-10-4-7-1-8(5-10)3-9(2-7)6-10/h7-9H,1-6H2 |
1-Fluoroadamantane - Physico-chemical Properties
| Molecular Formula | C10H15F |
| Molar Mass | 154.22 |
| Density | 1.06±0.1 g/cm3(Predicted) |
| Melting Point | 225 °C |
| Boling Point | 188.8±9.0 °C(Predicted) |
| Flash Point | 67.3°C |
| Vapor Presure | 0.813mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Almost white |
| Storage Condition | Room Temprature |
| Refractive Index | 1.494 |
1-Fluoroadamantane - Risk and Safety
| Hazard Symbols | Xi - Irritant |
1-Fluoroadamantane - Reference Information
| production method | 1. Preparation Method: polyhydrofluoropyridine (2): A dry ml polyethylene bottle was charged with 37.5g(0.475mol) of pyridine, and cooled in a dry ice-acetone bath until the pyridine solidified. Anhydrous hydrogen fluoride 87.5(4.37mol) was added and the reaction flask was vortexed to facilitate dissolution of the solid. After dissolution, it can be safely and slowly warmed to room temperature. Poly (hydrogen fluoride pyridine) (2). 1-fluoroadamantane (1): to a 250ml polyethylene reaction flask, 5G (0.033mol) of 1-adamantanol and 50ml of the above polyhydrofluoropyridine (2) solution were added, and the mixture was stirred at room temperature for 3 hours. 150ml of petroleum ether was added, and the reaction was vigorously stirred for 15min. The lower layer was separated by a polyethylene separatory funnel (the inorganic phase was destroyed by a large amount of water), and the organic matter was washed with 50ml of water, 50m of saturated sodium bicarbonate solution and 50ml of water in turn, dry over anhydrous magnesium sulfate. It is concentrated under reduced pressure to obtain 1-fluoroadamantane (1)4.5-4.6g, yield 88%-90%. It can be purified by sublimation or by recrystallization from methanol-carbon tetrachloride. Note: ① secondary alcohol and tertiary alcohol can be converted into fluorinated hydrocarbon with the yield of 70% ~ 90%. The polyhydrogen fluoride-pyridine reagent may directly cause an olefin, cyclopropane, a diazo compound, or the like to undergo a hydrofluorination addition reaction to generate an organic fluoride. [1] |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 1-FLUOROADAMANTANE Request for quotation
CAS: 768-92-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 768-92-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 1-Adamantyl Fluoride Visit Supplier Webpage Request for quotation
CAS: 768-92-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 768-92-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 1-FLUOROADAMANTANE Request for quotation
CAS: 768-92-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 768-92-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 1-Adamantyl Fluoride Visit Supplier Webpage Request for quotation
CAS: 768-92-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 768-92-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

