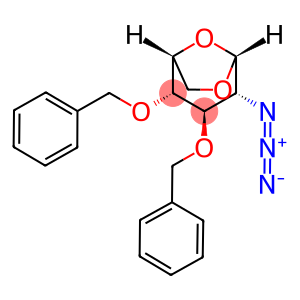
1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose
1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose
CAS: 55682-48-9
Molecular Formula: C20H21N3O4
1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose - Names and Identifiers
| Name | 1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose |
| Synonyms | 1,6-Anhydro-2-azido-3,4-di-O-benzyl-2-deoxy-β-D-glucopyranose β-D-Glucopyranose, 1,6-anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylMethyl)- 1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose (1R,2S,3R,4R,5R)-4-Azido-2,3-bis(benzyloxy)-6,8-dioxabicyclo[3.2. 1]octane (1R,2S,3R,4R,5R)-4-Azido-2,3-bis(benzyloxy)-6,8-dioxabicyclo[3.2.1]octane (non-preferred name) |
| CAS | 55682-48-9 |
| InChI | InChI=1/C20H21N3O4/c21-23-22-17-19(25-12-15-9-5-2-6-10-15)18(16-13-26-20(17)27-16)24-11-14-7-3-1-4-8-14/h1-10,16-20H,11-13H2/t16-,17-,18-,19-,20-/m1/s1 |
1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose - Physico-chemical Properties
| Molecular Formula | C20H21N3O4 |
| Molar Mass | 367.403 |
1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose - Introduction
1. 4-bis-O-(phenylmethyl)-beta-D-glucopyranose is an organic compound with the following properties:
1. Appearance: white solid
2. Solubility: Soluble in water and organic solvents, such as methanol and dimethylformamide
3. melting point: about 160-162 degrees Celsius
Its use is as follows:
1. Chemical synthesis intermediates: 1, 4-bis-O-(phenylmethyl)-beta-D-glucopyranose are commonly used as synthetic intermediates for drugs and bioactive molecules. It can be used as a substrate or as part of a reagent in organic synthesis reactions for the construction of complex organic molecules.
2. Drug research: The compound also has important applications in the field of drug research, and can be used to synthesize drugs with antiviral, anticancer and antibacterial activities.
The method of preparing 1, 4-bis-O-(phenylmethyl)-beta-D-glucopyranose generally includes the following steps:
1. First, the benzyl protecting group is introduced into the glucopyranose molecule to make it 3,4-bis-O-benzyl-beta-D-glucopyranose.
2. Under alkaline conditions, by reacting with nitrite, the amino group is introduced into the 2nd position of glucopyranose to form 2-azido-2-deoxy-3, 4-bis-O-Benzyl-beta-D-glucopyranose.
3. Finally, the hydroxyl group on the 6th position is removed by dehydration reaction to obtain 1, 4-bis-O-(phenylmethyl)-beta-D-glucopyranose.
Safety information regarding this compound should be carried out in strict accordance with safety procedures. Contact with skin and eyes should be avoided and personal protective equipment should be provided if necessary. In addition, during transportation and storage, care should be taken to avoid dangerous factors such as high temperature and exposure to fire sources.
1. Appearance: white solid
2. Solubility: Soluble in water and organic solvents, such as methanol and dimethylformamide
3. melting point: about 160-162 degrees Celsius
Its use is as follows:
1. Chemical synthesis intermediates: 1, 4-bis-O-(phenylmethyl)-beta-D-glucopyranose are commonly used as synthetic intermediates for drugs and bioactive molecules. It can be used as a substrate or as part of a reagent in organic synthesis reactions for the construction of complex organic molecules.
2. Drug research: The compound also has important applications in the field of drug research, and can be used to synthesize drugs with antiviral, anticancer and antibacterial activities.
The method of preparing 1, 4-bis-O-(phenylmethyl)-beta-D-glucopyranose generally includes the following steps:
1. First, the benzyl protecting group is introduced into the glucopyranose molecule to make it 3,4-bis-O-benzyl-beta-D-glucopyranose.
2. Under alkaline conditions, by reacting with nitrite, the amino group is introduced into the 2nd position of glucopyranose to form 2-azido-2-deoxy-3, 4-bis-O-Benzyl-beta-D-glucopyranose.
3. Finally, the hydroxyl group on the 6th position is removed by dehydration reaction to obtain 1, 4-bis-O-(phenylmethyl)-beta-D-glucopyranose.
Safety information regarding this compound should be carried out in strict accordance with safety procedures. Contact with skin and eyes should be avoided and personal protective equipment should be provided if necessary. In addition, during transportation and storage, care should be taken to avoid dangerous factors such as high temperature and exposure to fire sources.
Last Update:2024-04-09 21:54:55
Supplier List
Product Name: 1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose Request for quotation
CAS: 55682-48-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 55682-48-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 1,6-Anhydro-2-azido-2-deoxy-3,4-bis-O-(phenylmethyl)-beta-D-glucopyranose Request for quotation
CAS: 55682-48-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 55682-48-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
