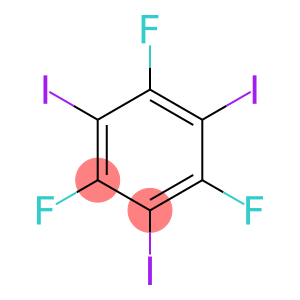
1,3,5-Trifluoro-2,4,6-triiodobenzene
1,3,5-Trifluoro-2,4,6-triiodobenzene
CAS: 84322-56-5
Molecular Formula: C6F3I3
1,3,5-Trifluoro-2,4,6-triiodobenzene - Names and Identifiers
1,3,5-Trifluoro-2,4,6-triiodobenzene - Physico-chemical Properties
| Molecular Formula | C6F3I3 |
| Molar Mass | 509.77 |
| Density | 3.029±0.06 g/cm3(Predicted) |
| Melting Point | 152℃ |
| Boling Point | 352.4±37.0 °C(Predicted) |
| Flash Point | 148.5°C |
| Water Solubility | Slightly soluble in water. |
| Vapor Presure | 7.82E-05mmHg at 25°C |
| Storage Condition | 2-8°C(protect from light) |
| Sensitive | Light Sensitive |
| Refractive Index | 1.698 |
1,3,5-Trifluoro-2,4,6-triiodobenzene - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Hazard Class | IRRITANT |
1,3,5-Trifluoro-2,4,6-triiodobenzene - Introduction
1,3,5-Trifluoro-2,4,6-triiodobenzene is an organic compound with the formula C6F3I3, which has the following properties:
1. Physical properties: 1,3,5-Trifluoro-2,4,6-triiodobenzene is a yellow crystal with a high melting point and boiling point. Solid at room temperature, soluble in organic solvents such as ethers and ketones, insoluble in water.
2. Chemical properties: The special structure of the compound makes it have certain chemical activity. It can be used as a reagent to participate in substitution reactions in organic synthesis and reactions.
3. Application: 1,3,5-Trifluoro-2,4,6-triiodobenzene has certain application value in the field of organic chemistry. It can be used as a reagent to participate in the substitution reactions of carbon-fluorine bonds and carbon-iodine bonds in organic reactions. In addition, it can also be used to study synthesis in fields such as material science and medicinal chemistry.
4. Preparation method: The preparation method of 1,3,5-Trifluoro-2,4,6-triiodobenzene is mainly achieved by substitution reaction. The common method is to prepare 2,4, 6-triiodobenzene first, and then carry out fluorination reaction under appropriate conditions to replace part of the iodine atoms of triiodobenzene with fluorine atoms.
5. Safety information: Since 1,3,5-Trifluoro-2,4,6-triiodobenzene is an organic compound, attention should be paid to its safety during use and storage. It may have irritating and harmful effects on the human body, so you should wear appropriate protective equipment and follow relevant safety procedures when operating. In addition, it may also cause pollution to the environment, and attention should be paid to prevent its leakage and discharge.
1. Physical properties: 1,3,5-Trifluoro-2,4,6-triiodobenzene is a yellow crystal with a high melting point and boiling point. Solid at room temperature, soluble in organic solvents such as ethers and ketones, insoluble in water.
2. Chemical properties: The special structure of the compound makes it have certain chemical activity. It can be used as a reagent to participate in substitution reactions in organic synthesis and reactions.
3. Application: 1,3,5-Trifluoro-2,4,6-triiodobenzene has certain application value in the field of organic chemistry. It can be used as a reagent to participate in the substitution reactions of carbon-fluorine bonds and carbon-iodine bonds in organic reactions. In addition, it can also be used to study synthesis in fields such as material science and medicinal chemistry.
4. Preparation method: The preparation method of 1,3,5-Trifluoro-2,4,6-triiodobenzene is mainly achieved by substitution reaction. The common method is to prepare 2,4, 6-triiodobenzene first, and then carry out fluorination reaction under appropriate conditions to replace part of the iodine atoms of triiodobenzene with fluorine atoms.
5. Safety information: Since 1,3,5-Trifluoro-2,4,6-triiodobenzene is an organic compound, attention should be paid to its safety during use and storage. It may have irritating and harmful effects on the human body, so you should wear appropriate protective equipment and follow relevant safety procedures when operating. In addition, it may also cause pollution to the environment, and attention should be paid to prevent its leakage and discharge.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 1,3,5-Trifluoro-2,4,6-triiodobenzene Visit Supplier Webpage Request for quotationCAS: 84322-56-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,3,5-Trifluoro-2,4,6-triiodobenzene Request for quotation
CAS: 84322-56-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 84322-56-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 1,3,5-Trifluoro-2,4,6-Triiodobenzene Visit Supplier Webpage Request for quotation
CAS: 84322-56-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 84322-56-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1,3,5-Trifluoro-2,4,6-triiodobenzene Visit Supplier Webpage Request for quotationCAS: 84322-56-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,3,5-Trifluoro-2,4,6-triiodobenzene Request for quotation
CAS: 84322-56-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 84322-56-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 1,3,5-Trifluoro-2,4,6-Triiodobenzene Visit Supplier Webpage Request for quotation
CAS: 84322-56-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 84322-56-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


