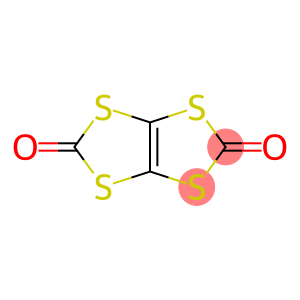
1,3,4,6-tetrathiapentalene-2,5-dione
1,3,4,6-tetrathiapentalene-2,5-dione
CAS: 64394-45-2
Molecular Formula: C4O2S4
1,3,4,6-tetrathiapentalene-2,5-dione - Names and Identifiers
1,3,4,6-tetrathiapentalene-2,5-dione - Physico-chemical Properties
| Molecular Formula | C4O2S4 |
| Molar Mass | 208.3 |
| Density | 2.05±0.1 g/cm3(Predicted) |
| Melting Point | 182°C (dec.)(lit.) |
| Boling Point | 393.0±52.0 °C(Predicted) |
| Flash Point | 194.7°C |
| Vapor Presure | 2.19E-06mmHg at 25°C |
| Appearance | powder to crystal |
| Color | Light orange to Yellow to Green |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.93 |
| MDL | MFCD00010403 |
1,3,4,6-tetrathiapentalene-2,5-dione - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. R42/43 - May cause sensitization by inhalation and skin contact. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S27 - Take off immediately all contaminated clothing. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| WGK Germany | 3 |
| HS Code | 29349990 |
1,3,4,6-tetrathiapentalene-2,5-dione - Introduction
1,3,4,6-tetrathiapentalene-2,5-dione is an organic compound with the chemical formula C4S4O2.
Nature:
1,3,4,6-tetrathiapentalene-2,5-dione is a solid, yellow crystal at room temperature. It has a relatively high melting and boiling point. It is an organic sulfur and organic oxygen heterocyclic compound, so it has some special chemical properties. It can undergo electron acceptor or donor reactions, and can undergo redox reactions.
Use:
1,3,4,6-tetrathiapentalene-2,5-dione has certain application value in the field of organic electronics and materials science. Due to its special electronic structure, it can be used as an organic semiconductor material for the construction of organic optoelectronic devices and organic field-effect transistors.
Preparation Method:
The preparation of 1,3,4,6-tetrathiapentalene-2,5-dione can be carried out by a synthetic route. One commonly used method is to react a para-dihalosulfide with a stable species (such as trifluoroacetic anhydride) under base catalysis to form 1,3, 4,6-tetrachlorothiazopentadiene. This is then reacted with hydrogen peroxide to give the 1,3,4,6-tetrathiapentalene-2,5-dione product.
Safety Information:
1,3,4,6-tetrathiapentalene-2,5-dione is generally stable under normal conditions of use, but it may cause irritation to the eyes, skin and respiratory system. Therefore, you should wear protective glasses, wear protective clothing, and ensure good ventilation. In addition, it should be stored in ionic gas and kept away from flammable substances.
Nature:
1,3,4,6-tetrathiapentalene-2,5-dione is a solid, yellow crystal at room temperature. It has a relatively high melting and boiling point. It is an organic sulfur and organic oxygen heterocyclic compound, so it has some special chemical properties. It can undergo electron acceptor or donor reactions, and can undergo redox reactions.
Use:
1,3,4,6-tetrathiapentalene-2,5-dione has certain application value in the field of organic electronics and materials science. Due to its special electronic structure, it can be used as an organic semiconductor material for the construction of organic optoelectronic devices and organic field-effect transistors.
Preparation Method:
The preparation of 1,3,4,6-tetrathiapentalene-2,5-dione can be carried out by a synthetic route. One commonly used method is to react a para-dihalosulfide with a stable species (such as trifluoroacetic anhydride) under base catalysis to form 1,3, 4,6-tetrachlorothiazopentadiene. This is then reacted with hydrogen peroxide to give the 1,3,4,6-tetrathiapentalene-2,5-dione product.
Safety Information:
1,3,4,6-tetrathiapentalene-2,5-dione is generally stable under normal conditions of use, but it may cause irritation to the eyes, skin and respiratory system. Therefore, you should wear protective glasses, wear protective clothing, and ensure good ventilation. In addition, it should be stored in ionic gas and kept away from flammable substances.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 1,3,4,6-TETRATHIAPENTALENE-2,5-DIONE Request for quotation
CAS: 64394-45-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 64394-45-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: [1,3]Dithiolo[4,5-d][1,3]dithiole-2,5-dione Visit Supplier Webpage Request for quotationCAS: 64394-45-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 1,3,4,6-Tetrathiapentalene-2,5-Dione Visit Supplier Webpage Request for quotationCAS: 64394-45-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 1,3,4,6-TETRATHIAPENTALENE-2,5-DIONE Request for quotation
CAS: 64394-45-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 64394-45-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: [1,3]Dithiolo[4,5-d][1,3]dithiole-2,5-dione Visit Supplier Webpage Request for quotationCAS: 64394-45-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 1,3,4,6-Tetrathiapentalene-2,5-Dione Visit Supplier Webpage Request for quotationCAS: 64394-45-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


