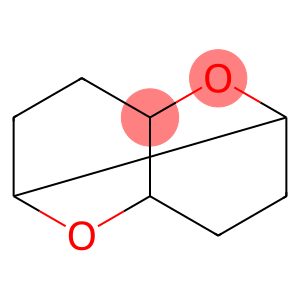
1,2,5,6-Diepoxycyclooctane
1,2,5,6-Diepoxycyclooctane
CAS: 27035-39-8
Molecular Formula: C8H12O2
1,2,5,6-Diepoxycyclooctane - Names and Identifiers
| Name | 1,2,5,6-Diepoxycyclooctane |
| Synonyms | 1,2,5,6-DIEPOXYCYCLOOCTANE 1,2,5,6-Diepoxycyclooctane 2,7-Dioxatricyclo(4.4.0.03,8)decane 2,7-dioxatricyclo[4.4.0.03,8]decane 5,10-dioxatricyclo[7.1.0.0~4,6~]decane (1R,4R,6R,9R)-5,10-dioxatricyclo[7.1.0.0~4,6~]decane (1R,4R,6R,9S)-5,10-dioxatricyclo[7.1.0.0~4,6~]decane (1R,4S,6S,9R)-5,10-dioxatricyclo[7.1.0.0~4,6~]decane |
| CAS | 27035-39-8 |
| EINECS | 248-180-1 |
| InChI | InChI=1/C8H12O2/c1-2-6-8(10-6)4-3-7-5(1)9-7/h5-8H,1-4H2/t5-,6+,7-,8+ |
1,2,5,6-Diepoxycyclooctane - Physico-chemical Properties
| Molecular Formula | C8H12O2 |
| Molar Mass | 140.18 |
| Density | 1.138 g/mL at 25 °C (lit.) |
| Boling Point | 70 °C/0.04 mmHg (lit.) |
| Flash Point | 221°F |
| Vapor Presure | 0.001mmHg at 25°C |
| Appearance | Liquid or Low Melting Solid |
| Color | Clear colorless to slightly yellow |
| Storage Condition | 2-8°C |
| Refractive Index | n20/D 1.496(lit.) |
1,2,5,6-Diepoxycyclooctane - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R68 - Possible risk of irreversible effects R40 - Limited evidence of a carcinogenic effect |
| Safety Description | 36/37 - Wear suitable protective clothing and gloves. |
| WGK Germany | 3 |
| HS Code | 29109000 |
1,2,5,6-Diepoxycyclooctane - Introduction
1,2,5,6-diepoxycycloctane is an important organic compound with the chemical formula C8H12O2. The following is a description of the properties, uses, preparation and safety information of the compound:
Nature:
-Cyclooctane is a colorless to light yellow liquid.
-It has a lower vapor pressure and a relatively high boiling point, and can exist in liquid form at room temperature.
-Cyclooctane is almost insoluble in water and soluble in organic solvents.
Use:
- 1,2, 5,6-diepoxycycloctane is widely used in resins, coatings, paints and preservatives.
-Due to the existence of its epoxy group, it can be used as a monomer of epoxy resin to produce various epoxy resin products.
-The epoxy group of cyclooctane can undergo ring-opening reactions with other compounds to produce a variety of organic functional compounds.
Method:
-Cyclooctane can be obtained by epoxidation of cyclooctane carbonate with an excess of an epoxidizing agent, such as formyl peroxide, tert-butyl peroxyformic acid, etc.
-The reaction is usually carried out under an inert atmosphere and at a low temperature to prevent unnecessary side reactions.
Safety Information:
-Cyclooctane is relatively stable under normal conditions of use, but may react violently at high temperatures, high pressures or in contact with strong oxidants.
-The compound is irritating to the skin and eyes, and should be rinsed with plenty of water immediately after contact.
-Wear protective gloves, glasses and protective clothing to ensure adequate ventilation when handling cyclooctane.
Please note that this is only a general introduction to 1,2, 5,6-diepoxycycloctane. The specific nature, use and safety information may need to be further studied according to the specific situation. When using chemicals, always follow the relevant safety procedures.
Nature:
-Cyclooctane is a colorless to light yellow liquid.
-It has a lower vapor pressure and a relatively high boiling point, and can exist in liquid form at room temperature.
-Cyclooctane is almost insoluble in water and soluble in organic solvents.
Use:
- 1,2, 5,6-diepoxycycloctane is widely used in resins, coatings, paints and preservatives.
-Due to the existence of its epoxy group, it can be used as a monomer of epoxy resin to produce various epoxy resin products.
-The epoxy group of cyclooctane can undergo ring-opening reactions with other compounds to produce a variety of organic functional compounds.
Method:
-Cyclooctane can be obtained by epoxidation of cyclooctane carbonate with an excess of an epoxidizing agent, such as formyl peroxide, tert-butyl peroxyformic acid, etc.
-The reaction is usually carried out under an inert atmosphere and at a low temperature to prevent unnecessary side reactions.
Safety Information:
-Cyclooctane is relatively stable under normal conditions of use, but may react violently at high temperatures, high pressures or in contact with strong oxidants.
-The compound is irritating to the skin and eyes, and should be rinsed with plenty of water immediately after contact.
-Wear protective gloves, glasses and protective clothing to ensure adequate ventilation when handling cyclooctane.
Please note that this is only a general introduction to 1,2, 5,6-diepoxycycloctane. The specific nature, use and safety information may need to be further studied according to the specific situation. When using chemicals, always follow the relevant safety procedures.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 1,2,5,6-DIEPOXYCYCLOOCTANE Request for quotation
CAS: 27035-39-8
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 27035-39-8
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 1,2,5,6-Diepoxycyclooctane Visit Supplier Webpage Request for quotation
CAS: 27035-39-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 27035-39-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 1,2,5,6-DIEPOXYCYCLOOCTANE Request for quotation
CAS: 27035-39-8
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 27035-39-8
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 1,2,5,6-Diepoxycyclooctane Visit Supplier Webpage Request for quotation
CAS: 27035-39-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 27035-39-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

