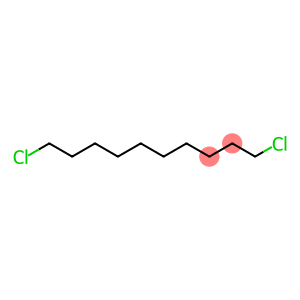
1,10-dichlorodecane
1,10-dichlorodecane
CAS: 2162-98-3
Molecular Formula: C10H20Cl2
1,10-dichlorodecane - Names and Identifiers
| Name | 1,10-dichlorodecane |
| Synonyms | 1,10-Dichlordecan 1,10-dichlorodecane 1,10-DICHLORODECANE 1,10-dichlorododecane DECAMETHYLENE CHLORIDE DECAMETHYLENE DICHLORIDE 1,10-DECAMETHYLENE DICHLORIDE 1,10-DICHLORODECANE FOR SYNTHESIS |
| CAS | 2162-98-3 |
| EINECS | 218-489-6 |
| InChI | InChI=1/C10H20Cl2/c11-9-7-5-3-1-2-4-6-8-10-12/h1-10H2 |
| InChIKey | RBBNTRDPSVZESY-UHFFFAOYSA-N |
1,10-dichlorodecane - Physico-chemical Properties
| Molecular Formula | C10H20Cl2 |
| Molar Mass | 211.17 |
| Density | 0.999 g/mL at 25 °C (lit.) |
| Melting Point | 15.6 °C (lit.) |
| Boling Point | 167-168 °C/28 mmHg (lit.) |
| Flash Point | 146 °C |
| Solubility | Chloroform, Methanol (Slightly) |
| Vapor Presure | 1.59-2.37Pa at 20-25℃ |
| Appearance | Liquid |
| Specific Gravity | 1 |
| Color | Clear colorless |
| Storage Condition | Sealed in dry,Room Temperature |
| Explosive Limit | 0.5%(V) |
| Refractive Index | 1.46-1.462 |
| Physical and Chemical Properties | Liquid. Melting point of 15.6 ℃, boiling point of 167-168 ℃(3.7kPa), relative density of 0.9945(20/4 ℃), refractive index of 1.4586. |
1,10-dichlorodecane - Risk and Safety
| Risk Codes | R38 - Irritating to the skin R36/37/38 - Irritating to eyes, respiratory system and skin. R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R53 - May cause long-term adverse effects in the aquatic environment |
| Safety Description | S28A - S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S60 - This material and its container must be disposed of as hazardous waste. |
| UN IDs | UN 3082 |
| WGK Germany | 3 |
| TSCA | Yes |
| HS Code | 29031980 |
| Hazard Class | 9 |
| Packing Group | III |
1,10-dichlorodecane - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| Introduction | Dichlorosilane, abbreviated as DCS, is an inorganic compound with the chemical formula H2SiCl2. |
| Properties | Dichlorosilane is hydrolyzed in water to produce polysiloxane (-H2Si-O-)n; and ammonia reacts ammonolysis to produce (-H2SiNH-)n[2]. |
| use | dichlorosilane can be used as the starting material of the semiconductor silicon layer in microelectronics. its advantage is that it can be decomposed at a lower temperature and has a higher silicon crystal growth rate. As a pharmaceutical intermediate. |
| Preparation | Dichlorosilane can be prepared by reacting silane and hydrogen chloride in the presence of aluminum trichloride catalyst in a stoichiometric ratio: it can also be prepared by trichlorosilane: 2 SiHCl3 SiH2Cl2 SiCl4 |
| Production method | It is obtained from chlorination of decanediol by thionyl chloride. Put decanediol and pyridine into the reaction pot, and add thionyl chloride dropwise under cooling. After adding, stir and react at room temperature for 3h, then reflux for 3h. Excess thionyl chloride was recovered and then distilled under reduced pressure to collect the 126-127°C (0.93kPa) fraction to obtain dichlorodecane. The yield was 88%. |
Last Update:2024-04-09 20:52:54
Supplier List
Product Name: 1,10-Dichlorodecane Request for quotation
CAS: 2162-98-3
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 2162-98-3
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Spot supply
Product Name: 1,10-Dichlorodecane Visit Supplier Webpage Request for quotationCAS: 2162-98-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,10-Dichlorodecane Request for quotation
CAS: 2162-98-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2162-98-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 1,10-Dichlorodecane Request for quotation
CAS: 2162-98-3
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 2162-98-3
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Product Name: 1,10-Dichlorodecane Request for quotation
CAS: 2162-98-3
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 2162-98-3
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Spot supply
Product Name: 1,10-Dichlorodecane Visit Supplier Webpage Request for quotationCAS: 2162-98-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,10-Dichlorodecane Request for quotation
CAS: 2162-98-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2162-98-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 1,10-Dichlorodecane Request for quotation
CAS: 2162-98-3
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 2162-98-3
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
View History



