(3S)-3-ethylmorpholine
(3S)-3-ethylmorpholine
CAS: 748117-01-3
Molecular Formula: C6H13NO
(3S)-3-ethylmorpholine - Names and Identifiers
| Name | (3S)-3-ethylmorpholine |
| Synonyms | (S)-3-Ethylmorpholine (3S)-3-Ethylmorpholine (3S)-3-ethylmorpholine Morpholine, 3-ethyl-,(3S)- morpholine, 3-ethyl-, (3S)- (3S)-3-Ethyl-morpholine HCl (S)-3-ethylmorpholine hydrochloride |
| CAS | 748117-01-3 |
| InChI | InChI=1/C6H13NO/c1-2-6-5-8-4-3-7-6/h6-7H,2-5H2,1H3/t6-/m0/s1 |
(3S)-3-ethylmorpholine - Physico-chemical Properties
| Molecular Formula | C6H13NO |
| Molar Mass | 115.17 |
| Density | 0.878g/cm3 |
| Boling Point | 160.6°C at 760 mmHg |
| Flash Point | 54.2°C |
| Vapor Presure | 2.36mmHg at 25°C |
| Storage Condition | 2-8℃ |
| Refractive Index | 1.413 |
(3S)-3-ethylmorpholine - Introduction
(3S)-3-ethylmorpholine is an organic compound with the chemical formula C7H15NO, also known as (3S)-3-ethylmorpholine. It is an amine compound commonly used in organic synthesis. The following is an introduction to the properties, uses, synthesis methods and safety information of (3S)-3-ethylmorpholine:
Nature:
(3S)-3-ethylmorpholine is a colorless liquid with a fishy odor. It is soluble in many organic solvents, such as ethanol, ether and benzene. At room temperature, its boiling point is about 137-139 degrees Celsius, and its density is about 0.89 g/ml.
Use:
(3S)-3-ethylmorpholine has a wide range of applications in the field of organic synthesis. It can be used as a catalyst, a reaction solvent, a ligand, and the like. It is often used in the synthesis of organic compounds, pharmaceutical intermediates, pigments and fragrances.
Preparation Method:
(3S)-3-ethylmorpholine can be prepared by a variety of methods. One commonly used method is the reaction of morpholine and ethyl bromide. The specific step is to react morpholine and ethyl bromide in a base medium to generate (3S)-3-ethylmorpholine. Higher purity products can be obtained by various purification methods.
Safety Information:
(3S)-3-ethylmorpholine is generally stable and safe under normal conditions of use. However, care should be taken to follow laboratory safety practices during use, including wearing appropriate protective equipment and ensuring good ventilation. Since it is toxic and may have adverse effects on the environment, the discharge of waste into the natural environment should be avoided. During the operation, care should be taken to prevent contact with the skin and eyes, and to avoid inhaling its vapor. In use and storage, please refer to the safety data form of the relevant chemical to ensure safe operation.
Nature:
(3S)-3-ethylmorpholine is a colorless liquid with a fishy odor. It is soluble in many organic solvents, such as ethanol, ether and benzene. At room temperature, its boiling point is about 137-139 degrees Celsius, and its density is about 0.89 g/ml.
Use:
(3S)-3-ethylmorpholine has a wide range of applications in the field of organic synthesis. It can be used as a catalyst, a reaction solvent, a ligand, and the like. It is often used in the synthesis of organic compounds, pharmaceutical intermediates, pigments and fragrances.
Preparation Method:
(3S)-3-ethylmorpholine can be prepared by a variety of methods. One commonly used method is the reaction of morpholine and ethyl bromide. The specific step is to react morpholine and ethyl bromide in a base medium to generate (3S)-3-ethylmorpholine. Higher purity products can be obtained by various purification methods.
Safety Information:
(3S)-3-ethylmorpholine is generally stable and safe under normal conditions of use. However, care should be taken to follow laboratory safety practices during use, including wearing appropriate protective equipment and ensuring good ventilation. Since it is toxic and may have adverse effects on the environment, the discharge of waste into the natural environment should be avoided. During the operation, care should be taken to prevent contact with the skin and eyes, and to avoid inhaling its vapor. In use and storage, please refer to the safety data form of the relevant chemical to ensure safe operation.
Last Update:2024-04-09 21:04:16
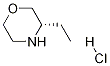
Supplier List
Spot supply
Product Name: (S)-3-Ethylmorpholine Visit Supplier Webpage Request for quotationCAS: 748117-01-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-3-Ethylmorpholine Request for quotation
CAS: 748117-01-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 748117-01-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: (S)-3-Ethylmorpholine Visit Supplier Webpage Request for quotationCAS: 748117-01-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-3-Ethylmorpholine Request for quotation
CAS: 748117-01-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 748117-01-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
View History

