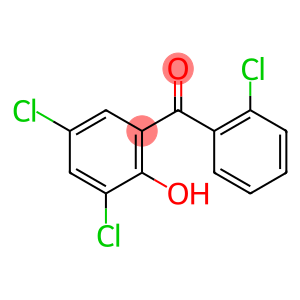
(2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)methanone
(2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)methanone
CAS: 99585-49-6
Molecular Formula: C13H7Cl3O2
(2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)methanone - Names and Identifiers
| Name | (2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)methanone |
| Synonyms | 3,5,2'-trichloro-2-hydroxy-benzophenone (2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)methanone Methanone, (2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)- |
| CAS | 99585-49-6 |
| InChI | InChI=1/C13H7Cl3O2/c14-7-5-9(13(18)11(16)6-7)12(17)8-3-1-2-4-10(8)15/h1-6,18H |
(2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)methanone - Physico-chemical Properties
| Molecular Formula | C13H7Cl3O2 |
| Molar Mass | 301.55 |
| Density | 1.494g/cm3 |
| Melting Point | 90-93°C(lit.) |
| Boling Point | 408.7°C at 760 mmHg |
| Flash Point | 201°C |
| Vapor Presure | 2.91E-07mmHg at 25°C |
| Refractive Index | 1.638 |
(2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)methanone - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
(2-chlorophenyl)(3,5-dichloro-2-hydroxyphenyl)methanone - Introduction
(2-chlorophenyl)(3,) is methanone, the chemical formula is C13H8Cl3O2, an organic compound. Its appearance is pale yellow to yellowish green crystalline, soluble in ethanol and dichloromethane, slightly soluble in water. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: light yellow to yellowish green crystal
-Molecular weight: 315.56g/mol
-Solubility: Soluble in ethanol and dichloromethane, slightly soluble in water
-melting point: 154-157 ℃
-Boiling point: 392.5 ℃
Use:
(2-chlorophenyl)(3, v) methanone is an important organic synthesis intermediate, which has a wide range of applications in chemical synthesis. It is often used as organic photosensitizer, dye synthesis intermediates, fluorescent dyes and pigment precursors.
Preparation Method:
a commonly used preparation method is to react 2,4-dinitrochlorobenzene with phthalic acid dianhydride to generate diketone compounds, and then to obtain (2-chlorophenyl)(3,) methanone through reduction, chlorination and other steps.
Safety Information:
- (2-chlorophenyl)(3) methanone need to be stored in a dry, cool place, away from fire and oxidizing agents.
-Wear appropriate personal protective equipment, including protective glasses and gloves.
-Avoid inhaling its dust or contact with skin, if accidental contact should be immediately washed with plenty of water.
-When used in a laboratory environment, follow safe operating procedures and correct waste disposal methods.
Nature:
-Appearance: light yellow to yellowish green crystal
-Molecular weight: 315.56g/mol
-Solubility: Soluble in ethanol and dichloromethane, slightly soluble in water
-melting point: 154-157 ℃
-Boiling point: 392.5 ℃
Use:
(2-chlorophenyl)(3, v) methanone is an important organic synthesis intermediate, which has a wide range of applications in chemical synthesis. It is often used as organic photosensitizer, dye synthesis intermediates, fluorescent dyes and pigment precursors.
Preparation Method:
a commonly used preparation method is to react 2,4-dinitrochlorobenzene with phthalic acid dianhydride to generate diketone compounds, and then to obtain (2-chlorophenyl)(3,) methanone through reduction, chlorination and other steps.
Safety Information:
- (2-chlorophenyl)(3) methanone need to be stored in a dry, cool place, away from fire and oxidizing agents.
-Wear appropriate personal protective equipment, including protective glasses and gloves.
-Avoid inhaling its dust or contact with skin, if accidental contact should be immediately washed with plenty of water.
-When used in a laboratory environment, follow safe operating procedures and correct waste disposal methods.
Last Update:2024-04-10 22:29:15